Introduction
Daratumumab is approved in many countries as monotherapy and in combination regimens for relapsed/refractory multiple myeloma (RRMM) and newly diagnosed multiple myeloma [1,2,3]. Daratumumab-based combinations have also demonstrated encouraging efficacy in lenalidomide-refractory RRMM [4, 5]. Carfilzomib is approved as monotherapy and in combination regimens, including daratumumab and dexamethasone (D-Kd), for RRMM [6]. In the phase 3 CANDOR study, D-Kd (intravenous [IV] daratumumab; carfilzomib 56 mg/m2 twice weekly) improved progression-free survival (PFS) versus Kd in the overall population and lenalidomide-refractory patients [7, 8]. In the phase 3 A.R.R.O.W. study, once-weekly carfilzomib (70 mg/m2) significantly prolonged PFS versus twice-weekly carfilzomib (27 mg/m2), providing a safe and more convenient Kd dosing regimen [9].
Preliminary results from the phase 2 PLEIADES study (median follow-up, 9.2 months) and the phase 1b EQUULEUS study (median follow-up, 16.6 months) showed that combining subcutaneous daratumumab (DARA SC) and daratumumab IV, respectively, with Kd (carfilzomib 70 mg/m2 weekly) was well tolerated and induced deep responses in RRMM patients [10, 11]. We report final data from PLEIADES and EQUULEUS, with a median follow-up of 12.4 and 23.7 months, respectively.
Methods
PLEIADES (ClinicalTrials.gov Identifier: NCT03412565) evaluated DARA SC (daratumumab 1800 mg co-formulated with recombinant human hyaluronidase PH20 [rHuPH20; 2000 U/mL; ENHANZE® drug delivery technology, Halozyme, Inc., San Diego, CA, USA]) plus weekly Kd (carfilzomib 70 mg/m2; dexamethasone 40 mg) in RRMM patients with 1 prior line of lenalidomide-based therapy. EQUULEUS (NCT01998971) evaluated daratumumab IV plus weekly Kd (carfilzomib 70 mg/m2; dexamethasone 40 mg) in RRMM patients after 1 to 3 prior lines of therapy (including bortezomib and an immunomodulatory drug). Patients in both studies received 28-day cycles of D-Kd until disease progression. Primary endpoints were the overall response rate (ORR) in PLEIADES and safety and tolerability in EQUULEUS. Supplementary Information includes additional methodology.
Results
A total of 66 and 85 patients received D-Kd in PLEIADES and EQUULEUS, respectively. Patient demographic and baseline characteristics are summarized in Table S1. In PLEIADES and EQUULEUS, the median (range) age was 61 (42–84) and 66 (38–85) years, respectively, 16/44 (36.4%) and 13/67 (19.4%) patients had high-risk cytogenetics, and 41 (62.1%) and 51 (60.0%) were lenalidomide-refractory. Patients in PLEIADES and EQUULEUS had received a median (range) of 1 (1–1) and 2 (1–4) prior lines of therapy, respectively, and 60 (90.9%) and 85 (100%) patients had received prior proteasome inhibitor therapy. In PLEIADES and EQUULEUS, 31 (47.0%) and 50 (58.8%) patients, respectively, discontinued treatment, primarily due to progressive disease. Median (range) treatment duration was 12.0 (0–21) months in PLEIADES and 19.8 (0.3–34.5) months in EQUULEUS. Patient disposition and drug exposure are further described in Supplementary Information.
Pharmacokinetic data are provided for pharmacokinetic-evaluable populations (PLEIADES, n = 65; EQUULEUS, n = 85 [single first dose, n = 10; split first dose, n = 75]). The maximum concentration of DARA SC was observed on Cycle 1, Day 4 after the first dose or Cycle 3, Day 4 after the ninth dose. For daratumumab IV, the maximum concentration was observed on Cycle 1, Day 1 (end of infusion) after the first dose or Cycle 3, Day 1 (end of infusion) after the ninth dose. In both studies, serum trough concentration (Ctrough) increased to maximum Ctrough on Cycle 3, Day 1 pre-dose, then decreased with less frequent dosing. DARA SC provided numerically higher Ctrough (within a similar range) versus daratumumab IV. Subgroup analysis of serum daratumumab concentration based on body weight in PLEIADES is presented in Table S2. In EQUULEUS, pharmacokinetic profiles of single and split first daratumumab doses were similar from Cycle 2, Day 1 pre-infusion onward (Table S3). No patient in the immunogenicity-evaluable population of either study tested positive for anti-daratumumab antibodies. In PLEIADES, 3/64 (4.7%) patients in the rHuPH20 immunogenicity-evaluable population had treatment-emergent anti-rHuPH20 antibodies after DARA SC administration; none were neutralizing.
The most common any-grade and grade 3/4 treatment-emergent adverse events (TEAEs) in PLEIADES are summarized in Table 1. Grade 3/4 infections occurred in 9 (13.6%) patients, most commonly pneumonia (4.5%). Serious TEAEs were reported in 22 (33.3%) patients, most commonly pneumonia (4.5%). One (1.5%) patient discontinued treatment due to a TEAE. Three patients had grade 5 TEAEs: 1 each with COVID-19 pneumonia, sepsis, and respiratory failure.
The most common any-grade and grade 3/4 TEAEs in EQUULEUS are summarized in Table 1. Grade 3/4 infections were reported in 18 (21.2%) patients, most commonly pneumonia (4.7%). Serious TEAEs occurred in 41 (48.2%) patients, most commonly basal cell carcinoma, pneumonia, and upper respiratory tract infection (4.7% each). Five (5.9%) patients discontinued treatment due to TEAEs. Three patients had grade 5 TEAEs: two with general physical health deterioration and one with multiple organ dysfunction syndrome.
Two (3.0%) patients in PLEIADES had grade 3/4 cardiac TEAEs: one each with grade 3 cardiac failure and grade 4 left ventricular dysfunction. In EQUULEUS, 9 (10.6%) patients had grade 3/4 cardiac TEAEs, including sinus tachycardia, cardiac failure, systolic dysfunction (n = 2 each) and atrial fibrillation, congestive cardiomyopathy, left ventricular failure, myocardial ischemia, and myocarditis (n = 1 each). In both studies, the median left ventricular ejection fraction did not notably change over time from baseline. Additional details are included in Supplementary Information.
Three (4.5%) patients had infusion-related reactions (IRRs) with DARA SC in PLEIADES; two had grade 3 IRRs. All patients with IRRs experienced them on the first administration; the median (range) time to IRR onset was 65 (4–75) min. Local injection-site reactions occurred in 7 (10.6%) patients; all were grade 1/2. In EQUULEUS, 6 (60.0%) patients who received a single first dose and 31 (41.3%) who received a split first dose had IRRs with daratumumab IV. Most IRRs were mild (2 patients had grade 3/4 IRRs) and occurred during the first infusion. Five (50.0%) patients experienced IRRs during Cycle 1, Day 1 with a single first daratumumab dose; 27 (36.0%) experienced IRRs during Cycle 1, Day 1 with a split first daratumumab dose.
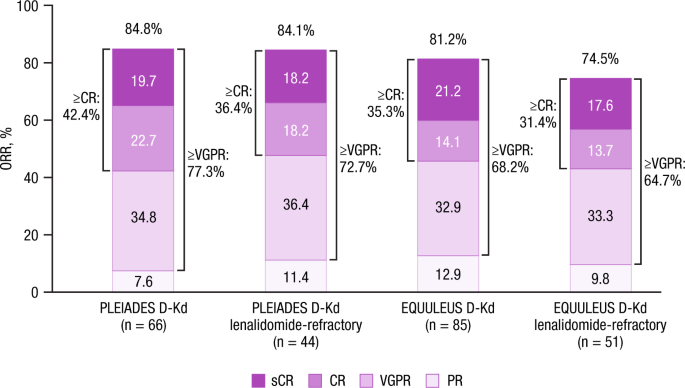
In PLEIADES (median [range] follow-up, 12.4 [0.2–20.6] months), ORR was 84.8% in the all-treated population and 84.1% in lenalidomide-refractory patients (Fig. 1). ORR was 75.0% (12/16) and 82.1% (23/28) in patients with high and standard cytogenetic risk, respectively. The median duration of response was not reached; the 9-month duration of response rate was 85.4%. PFS and overall survival (OS) were not analyzed.
Data were shown for the all-treated population. Note: Due to rounding, percentages may not add up to the total percentage for each category. CR complete response, D-Kd daratumumab/carfilzomib/dexamethasone, ORR overall response rate, PR partial response, sCR stringent complete response, VGPR very good partial response.
In EQUULEUS (median [range] follow-up, 23.7 [0.5–34.7] months), ORR was 81.2% in the all-treated population and 74.5% in lenalidomide-refractory patients (Fig. 1). The median duration of response was 27.5 months; the 9-month duration of response rate was 88.3%. Median PFS was 25.7 months in the all-treated population (Fig. S1A) and 22.3 months in lenalidomide-refractory patients; estimated 24-month PFS rates were 52.7% and 46.9%, respectively. The median time to subsequent anti-cancer therapy was 29.2 months. Median OS was not reached; the estimated 24-month OS rate was 71.2% (Fig. S1B).
Discussion
With additional follow-up in PLEIADES and EQUULEUS, no new safety concerns were identified, and the overall safety profile of D-Kd was consistent between studies. As previously demonstrated, lower IRR rates and shorter administration times were observed with the DARA SC–based versus daratumumab IV–based regimen. In EQUULEUS, split first daratumumab dosing produced similar safety and pharmacokinetic profiles as a single first dose from Cycle 2, Day 1 pre-infusion onward and decreased median infusion duration on Cycle 1, Day 1, which may improve patient convenience. Following weekly dosing, Ctrough of daratumumab at Cycle 3, Day 1 pre-dose was numerically higher (within a similar range) with DARA SC versus daratumumab IV. DARA SC administration achieved adequate and consistent exposure for all body weight subgroups. The incidence of treatment-emergent anti-daratumumab antibodies was 0% in both studies, indicating a low risk of immunogenicity to D-Kd. The incidence of treatment-emergent rHuPH20 antibodies was consistent with other DARA SC studies [12, 13].
Final analysis results from both studies showed D-Kd consistently induced deep responses regardless of lenalidomide refractoriness. ORRs in PLEIADES and EQUULEUS were comparable (84.8% and 81.2%, respectively). Encouraging 24-month PFS and OS rates of 52.7 and 71.2%, respectively, were demonstrated in EQUULEUS.
Cross-trial comparisons should be interpreted cautiously due to study differences. The phase 3 CANDOR study [8] compared daratumumab IV plus Kd versus Kd in RRMM patients with 1 to 3 prior lines of therapy. Patients in CANDOR received carfilzomib 56 mg/m2 twice weekly (monthly cumulative dose, 336 mg/m2 [Cycle 1, 264 mg/m2]), while patients in PLEIADES and EQUULEUS received carfilzomib 70 mg/m2 weekly (monthly cumulative dose, 210 mg/m2 [Cycle 1, 160 mg/m2]; ie, 62.5% of CANDOR monthly dose). A higher proportion of patients in PLEIADES and EQUULEUS had prior lenalidomide exposure (100% and 95%, respectively) or were refractory to lenalidomide (62% and 60%) versus the CANDOR D-Kd arm (39% and 32%, respectively). ORRs in PLEIADES (84.8%) and EQUULEUS (81.2%) were comparable to that reported at a median follow-up of 27.8 months in the CANDOR D-Kd arm (84%), with higher complete response or better rates (42.4% and 35.3% vs 33%, respectively). Median PFS was similar between the D-Kd arms of EQUULEUS and CANDOR (25.7 and 28.6 months, respectively) [8]. Overall, D-Kd demonstrated efficacy in PLEIADES, EQUULEUS, and CANDOR, supporting its use for RRMM, including lenalidomide-refractory multiple myeloma. Safety profiles were generally similar with D-Kd utilizing weekly carfilzomib dosing in PLEIADES and EQUULEUS and twice-weekly carfilzomib dosing in CANDOR; however, the incidence of fatal TEAEs was lower in PLEIADES (3/66 [4.5%]) and EQUULEUS (3/85 [3.5%]) compared to the CANDOR D-Kd arm (27/308 [8.8%]) [8]. Results from the phase 3 A.R.R.O.W. study also reinforce the favorable benefit-risk profile for once-weekly carfilzomib dosing compared with a twice-weekly treatment schedule [9]. Once-weekly carfilzomib dosing may improve cost-effectiveness and convenience for patients and healthcare providers [14].
Limitations of PLEIADES and EQUULEUS include small sample sizes and lack of comparator arms. Also, patients in PLEIADES were only followed for up to 8 weeks after the last study treatment; therefore, PFS and OS were not evaluated.
In conclusion, D-Kd continues to be well tolerated and effective in RRMM patients, including lenalidomide-refractory patients. Final analysis results from PLEIADES and EQUULEUS further support D-Kd as a standard treatment regimen for RRMM.
Data availability
The data sharing policy of Janssen Pharmaceutical Companies of Johnson & Johnson is available at https://www.janssen.com/clinical-trials/transparency. As noted on this site, requests for access to the study data can be submitted through Yale Open Data Access (YODA) Project site at http://yoda.yale.edu.
References
DARZALEX® (daratumumab) injection, for intravenous use [package insert]. Janssen Biotech, Inc. 2022.
European Medicines Agency. DARZALEX 20 mg/mL concentrate for solution for infusion [summary of product characteristics]. https://www.ema.europa.eu/en/documents/product-information/darzalex-epar-product-information_en.pdf. Accessed Oct 2022.
DARZALEX FASPRO® (daratumumab and hyaluronidase-fihj) injection, for subcutaneous use [package insert]. Janssen Biotech, Inc. 2022.
Dimopoulos MA, Terpos E, Boccadoro M, Delimpasi S, Beksac M, Katodritou E, et al. APOLLO Trial Investigators. Daratumumab plus pomalidomide and dexamethasone versus pomalidomide and dexamethasone alone in previously treated multiple myeloma (APOLLO): an open-label, randomised, phase 3 trial. Lancet Oncol. 2021;22:801–12.
Mateos MV, Sonneveld P, Hungria V, Nooka AK, Estell JA, Barreto W, et al. Daratumumab, bortezomib, and dexamethasone versus bortezomib and dexamethasone in patients with previously treated multiple myeloma: three-year follow-up of CASTOR. Clin Lymphoma Myeloma Leuk. 2020;20:509–18.
KYPROLIS® (carfilzomib) for injection, for intravenous use [package insert]. Onyx Pharmaceuticals, Inc. 2021.
Dimopoulos M, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): results from a randomised, multicentre, open-label, phase 3 study. Lancet. 2020;396:186–97.
Usmani SZ, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): updated outcomes from a randomised, multicentre, open-label, phase 3 study. Lancet Oncol. 2022;23:65–76.
Moreau P, Mateos MV, Berenson JR, Weisel K, Lazzaro A, Song K, et al. Once weekly versus twice weekly carfilzomib dosing in patients with relapsed and refractory multiple myeloma (A.R.R.O.W.): interim analysis results of a randomised, phase 3 study. Lancet Oncol. 2018;19:953–64.
Moreau P, Chari A, Haenel M, Oriol A, Rodriguez-Otero P, McCarthy H, et al. Subcutaneous daratumumab (DARA SC) plus standard-of-care (SoC) regimens in multiple myeloma (MM) across lines of therapy in the phase 2 PLEIADES study: initial results of the DARA SC plus carfilzomib/dexamethasone (D-Kd) cohort, and updated results for the DARA SC plus bortezomib/melphalan/prednisone (D-VMP) and DARA SC plus lenalidomide/dexamethasone (D-Rd) cohorts. Blood. 2020;136:28–30.
Chari A, Martinez-Lopez J, Mateos MV, Bladé J, Benboubker L, Oriol A, et al. Daratumumab plus carfilzomib and dexamethasone in patients with relapsed or refractory multiple myeloma. Blood. 2019;134:421–31.
San-Miguel J, Usmani SZ, Mateos MV, van de Donk NWCJ, Kaufman JL, Moreau P, et al. Subcutaneous daratumumab in patients with relapsed or refractory multiple myeloma: Part 2 of the open-label, multicenter, dose-escalation phase 1b study (PAVO). Haematologica. 2021;106:1725–32.
Mateos MV, Nahi H, Legiec W, Grosicki S, Vorobyev V, Spicka I. et al. Subcutaneous versus intravenous daratumumab in patients with relapsed or refractory multiple myeloma (COLUMBA): a multicentre, open-label, non-inferiority, randomised, phase 3 trial. Lancet Haematol. 2020;7:e370–80.
Kumar SK, Majer I, Panjabi S, Medhekar R, Campioni M, Dimopoulos MA. Cost-effectiveness of once weekly carfilzomib 70 mg/m2 plus dexamethasone in patients with relapsed and refractory multiple myeloma in the United States. Expert Rev Hematol. 2020;13:687–96.
Acknowledgements
These studies (ClinicalTrials.gov Identifiers: NCT03412565 and NCT01998971) were sponsored by Janssen Research & Development, LLC. We thank the patients participating in the PLEIADES and EQUULEUS studies and their families, the staff members at the study sites, the data and safety monitoring committees, and the staff members who were involved in the data collection and analyses. We also thank Amgen for supplying the carfilzomib used in these studies. Medical writing and editorial support were provided by Lisa Shannon, PharmD, of Lumanity Communications Inc., and were funded by Janssen Global Services, LLC.
Author information
Authors and Affiliations
Contributions
All authors interpreted the data, reviewed the manuscript, approved the final version, decided to publish this report, and vouch for the data accuracy and completeness.
Corresponding author
Ethics declarations
Competing interests
PM received honoraria from Bristol Myers Squibb/Celgene, Janssen-Cilag, AbbVie, Amgen, Takeda, Oncopeptides, Sanofi, and GlaxoSmithKline. AC served as a consultant or in an advisory role for Amgen, Janssen Oncology, Seattle Genetics, Karyopharm Therapeutics, Genzyme, Oncopeptides, Takeda, Antengene, GlaxoSmithKline, Secura Bio, Shattuck Labs, Genentech, AbbVie, and Bristol Myers Squibb/Celgene; and received research funding from Celgene, Janssen, Amgen, Seattle Genetics, Takeda, and Pharmacyclics. AO served on advisory boards for Sanofi, GlaxoSmithKline, Bristol Myers Squibb, Amgen, and Janssen. JM-L consulted for, received honoraria from, and served on a board of directors or advisory committees for Janssen; consulted for and received research funding from Bristol Myers Squibb and Incyte; and consulted for Novartis. MH received honoraria from Amgen, Novartis, Roche, Celgene, Takeda, and Bayer. CT received honoraria from and served in a consulting or advisory role for Janssen, Celgene, Takeda, Amgen, and AbbVie; and had travel, accommodations, or other expenses paid or reimbursed by Janssen, Takeda, Amgen, and AbbVie. SA served as a consultant for GlaxoSmithKline, Sanofi, Bristol Myers Squibb, Takeda, BeiGene, Pharmacyclics, Amgen, and Janssen; and received research funding from GlaxoSmithKline, Bristol Myers Squibb, Pharmacyclics, Amgen, Janssen, Cellectar, Xencor, and AbbVie. BB received honoraria from Janssen, Amgen, Takeda, and Sanofi. JdlRC received honoraria from Bristol Myers Squibb/Celgene, Jansen-Cilag, Amgen, Takeda, Sanofi, and GlaxoSmithKline; and received travel support from Janssen-Cilag. CE received honoraria from Bristol Myers Squibb/Celgene, Janssen-Cilag, Amgen, Sanofi, and GlaxoSmithKline. M-VM received honoraria derived from lectures and participation in advisory boards from Janssen, Celgene, Takeda, Amgen, GlaxoSmithKline, AbbVie, Pfizer, Regeneron, Adaptive Biotechnologies, Roche, and Seattle Genentech. HS received honoraria from Bristol Myers Squibb/Celgene, Janssen-Cilag, AbbVie, Amgen, Takeda, Oncopeptides, Sanofi, and GlaxoSmithKline; and received travel support from Bristol Myers Squibb/Celgene, Janssen-Cilag, AbbVie, Amgen, Sanofi, and GlaxoSmithKline. PR-O participated in speakers bureaus for and received honoraria from Bristol Myers Squibb/Celgene, Janssen, Amgen, GlaxoSmithKline, Sanofi, Regeneron, and Oncopeptides. CH received honoraria from Janssen, Bristol Myers Squibb, Amgen, Takeda, and AbbVie. LK served on advisory boards for and/or received honoraria or travel support from Celgene/Bristol Myers Squibb, Janssen, Takeda, Amgen, and GlaxoSmithKline. ASB served as a consultant for, received honoraria from, and participated in speakers bureaus for Takeda. LR received honoraria from Janssen, Celgene, Amgen, Takeda, Sanofi, and GlaxoSmithKline. HT served on an advisory board for Bristol Myers Squibb; and participated in speakers bureaus for ADP. SY, JW, IN, MQ, and MK are employees of Janssen Research & Development, LLC, and may hold equity. MD was employed by Janssen Research & Development, LLC, at the time of the study. HG received grants and/or provision of Investigational Medicinal Products from Amgen, Bristol Myers Squibb, Celgene, Chugai, Dietmar Hopp Foundation, Janssen, Johns Hopkins University, and Sanofi; received research support from Amgen, Bristol Myers Squibb, Celgene, Chugai, Janssen, Incyte, Molecular Partners, Merck Sharp & Dohme, Sanofi, Mundipharma GmbH, Takeda, and Novartis; served on advisory boards for Adaptive Biotechnologies, Amgen, Bristol Myers Squibb, Celgene, Janssen, Sanofi, and Takeda; and received honoraria from Amgen, Bristol Myers Squibb, Celgene, Chugai, GlaxoSmithKline, Janssen, Novartis, and Sanofi. JB, LB, and ST have nothing to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Moreau, P., Chari, A., Oriol, A. et al. Daratumumab, carfilzomib, and dexamethasone in relapsed or refractory myeloma: final analysis of PLEIADES and EQUULEUS. Blood Cancer J. 13, 33 (2023). https://doi.org/10.1038/s41408-023-00805-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41408-023-00805-x