氯化钇
(重定 向 自 三 氯化钇)
| 氯化钇 | |
|---|---|
| |
| IUPAC 氯化钇(III) Yttrium(III) chloride | |
| 识别 | |
| CAS |
10361-92-9 |
| ChemSpider | 59696 |
| SMILES |
|
| InChI |
|
| InChIKey | PCMOZDDGXKIOLL-DFZHHIFOAW |
| RTECS | ZG3150000 |
| YCl3 | |
| 195.26 g·mol⁻¹ | |
| 2.67 g/cm3 | |
| 熔点 | 721 °C |
| 1507 °C[1] | |
| 82 g/100 mL | |
| 60.1 g/100 mL 60.6 g/100 mL吡啶 (15°C)[2] | |
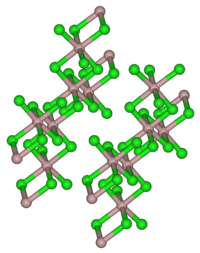
| 结构 | |
| Monoclinic, mS16 | |
| C12/m1, No. 12 | |
| 危险 | |
| 闪点 | |
| 其他阴离 |
氧化钇 |
| 其他阳离 |
氯化钪 氯化镧 氯化锕 |
氯化钇
结构
[编辑]制 备和反 应
[编辑]无水氯化钇
- 10 NH4Cl + Y2O3 → 2 (NH4)2[YCl5] + 6 NH3 + 3 H2O
- YCl3·6H2O + 2 NH4Cl → (NH4)2[YCl5] + 6 H2O
- (NH4)2[YCl5] → 2 NH4Cl + YCl3
如果
参考 资料
[编辑]- ^
Yttrium & Compounds, United States Occupational Safety and Health Administration, 2007-01-11 [2008-05-29], (
原始 内容 存 档于2013-03-02) - ^ Spencer, James F., The Metals of the Rare Earths, New York: Longmans, Green, and Co: 135, 1919 [2008-05-29]
- ^ , Meyer, G. The Ammonium Chloride Route to Anhydrous Rare Earth Chlorides-The Example of YCl3. Inorganic Syntheses. 1989, 25: 146–150. ISBN 978-0-470-13256-2. doi:10.1002/9780470132562.ch35.
- ^ Edelmann, F. T.; Poremba, P. Herrmann, W. A. (ed.) , 编. Synthetic Methods of Organometallic and Inorganic Chemistry VI. Stuttgart: Georg Thieme Verlag. 1997. ISBN 3-13-103021-6.
- ^ Taylor, M.D.; Carter, C.P. Preparation of anhydrous lanthanide halides, especially iodides. Journal of Inorganic and Nuclear Chemistry: 387–391. doi:10.1016/0022-1902(62)80034-7.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
