砷酸钠
| 砷酸钠 | |
|---|---|
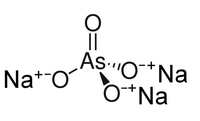
| |
| IUPAC Sodium dihydrogen arsenate | |
| 别名 | sodium arsenate |
| 识别 | |
| CAS |
13464-38-5(无水) 13510-46-8( |
| PubChem | 47275 23622126 57348098 |
| ChemSpider | 43027, 29325835 |
| SMILES |
|
| InChI |
|
| InChIKey | CDBAKLRDFBGJOX-UHFFFAOYSA-K |
| ChEBI | 84070 |
| Na3AsO4 | |
| 207.89 g·mol⁻¹ | |
| 无色 | |
| 1.517 g/cm3 ( | |
| 熔点 | 995—1,005 °C(1,268—1,278 K)(无水)[1] 60 °C(333 K)( |
| 危险 | |
GHS危险  
| |
| GHS |
危险 |
| H-术语 | H301, H331, H350, H410 |
| P-术语 | P201, P202, P261, P264, P270, P271, P273, P281, P301+310, P304+340, P308+313, P311, P321, P330 |
砷酸钠
制 备
[编辑]砷酸钠可
- H3AsO4 + 3 NaOH → Na3AsO4 + 3 H2O
- 2 NiAs + 7 NaNO3 + 6 NaOH → 2 NiO + 2 Na3AsO4 + 7 NaNO2 + 3 H2O
性 质
[编辑]砷酸钠和
参考 文献
[编辑]- ^ 1.0 1.1 Buketov, E. A.; Polukarov, A. N.; Isabaev, S. M.; Zhambekov, M. I. Thermal stability of sodium arsenates. Vestnik Akademii Nauk Kazakhskoi SSR, 1973. 29 (6): 30-33. ISSN: 0002-3213.
- ^ Sagadieva, A. K.; Makhmetov, M. Zh.; Kozorin, L. G. Crystal hydrates of sodium arsenate (Na3AsO4). Zhurnal Neorganicheskoi Khimii, 1980. 25 (4): 1129-1132. ISSN: 0044-457X.
- ^ Chavchanidze, N. G.; Giuashvili, I. I.; Ugulava, M. M.; Gigauri, R. D. Preparation of alkali metal arsenates. Zhurnal Neorganicheskoi Khimii, 1990. 35 (4): 1074-1076. ISSN: 0044-457X.
- ^ Fester, G. A. Sodium arsenate from nickel arsenides. Rev. fac. ing. quim., 1956. 25: 143-151. CAN52: 10344.
- ^ Kekenadze, M.; Rusia, M.; Kutsia, L.; Dzotsenidze, Z.; Gigauri, R. Synthesis and study of ammines of d-metals' (III) tetraoxoarsenates. Bulletin of the Georgian Academy of Sciences, 2000. 162 (2): 265-268. ISSN: 1560-0262.
- ^ Bugenov, E. S.; Savel'ev, A. E. Mechanism of homogeneous reduction of arsenate ions with Formalin. Kompleksnoe Ispol'zovanie Mineral'nogo Syr'ya, 1979. 4: 22-28. ISSN: 0202-1382.
| ||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
