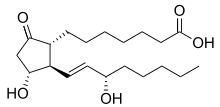
Prostaglandin E1 (PGE1) is a naturally occurring prostaglandin and is also used as a medication (alprostadil).[2]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Caverject, Muse, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a695022 |
| License data |
|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.010.925 |
| Chemical and physical data | |
| Formula | C20H34O5 |
| Molar mass | 354.487 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
In infants with congenital heart defects, it is delivered by slow injection into a vein to open the ductus arteriosus until surgery can be carried out.[3] By injection into the penis or placement in the urethra, it is used to treat erectile dysfunction.[4]
Common side effects when given to babies include decreased breathing, fever, and low blood pressure.[2] When injected into the penis for erectile dysfunction; side effects may include penile pain, bleeding at the site of injection, and prolonged erection (priapism).[2] Prostaglandin E1 is in the vasodilator family of medications.[2] It works by opening blood vessels and relaxing smooth muscle.[2]
Prostaglandin E1 was isolated in 1957 and approved for medical use in the United States in 1981.[2][5] It is on the World Health Organization's List of Essential Medicines.[6]
Biosynthesis edit
Prostaglandin E1 is biosynthesized on an as-needed basis from dihomo-
Medical uses edit
Patent ductus arteriosus edit
Alprostadil is also used in maintaining a patent ductus arteriosus in newborns, primarily for an infant with ductal-dependent congenital heart disease, including cyanotic lesions and acyanotic lesions.
Sexual dysfunction edit
Alprostadil is sold in the United States as urethral suppositories (brand name Muse[8]) and in injectable form (brand names: Edex[9] and Caverject[10]). As a penile suppository, the medication is inserted into the urethra at least ten minutes before the erection is needed. Other forms similarly fast-acting, but instead are injected by the syringe directly into the corpus cavernosum of the penis.
Alprostadil is also available as a generic. It must be mixed by a compounding pharmacy. The different formulations, including Bimix and Trimix, may include papaverine and/or phentolamine. A typical mix might be 30 mg of papaverine, 2 mg of phentolamine, and 20
This section contains content that is written like an advertisement. (May 2021) |
The compound has been made into an applicable topical cream form known as Vitaros,[11] made by Takeda UK Ltd., it contains either 200 or 300
Off-brand Uses, Interactions edit
Clinical trials for the treatment showed positive results in around 3,000 men that it was tested on; it is said to be usable by men with diabetes or heart problems and those who have undergone a prostatectomy.[12] It has no known interactions with food, alcohol or other medications.
Misoprostol is another synthetic prostaglandin E1 analog used to prevent gastric ulcers when taken on a continuous basis,[13] to treat missed miscarriage,[14] to induce labor,[15] and to induce abortion.[16]
Critical limb ischemia edit
Prostanoids, including alprostadil, do not reduce the risk of limb amputation but may offer a slight improvement in rest-pain and leg ulcer healing in persons with critical limb ischemia.[17]
Contrast-induced nephropathy edit
Preventative administration of alprostadil may reduce the risk of kidney injury (specifically contrast-induced nephropathy) in persons having cardiac angiography or percutaneous coronary intervention.[18][19]
Adverse effects edit
- Accidental injury (Muse only)
- Apnea
- Bleeding:
- Cerebral
- Urethral
- Bradycardia
- Cardiac arrest
- Congestive heart failure
- Cortical proliferation of long bones
- Diarrhea
- Disseminated intravascular coagulation
- Edema
- Fever
- Flushing
- Hyperemia
- Hypotension
- Injection-site haematoma
- Injection-site ecchymosis (Caverject only)
- Pain:
- Back
- Pelvic
- Penile
- Testicular (Muse only)
- Urethral
- Prolonged erection
- Penile fibrosis
- Second-degree heart block
- Seizures
- Sepsis
- Shock
- Spasm of right ventricle infundibulum
- Supraventricular tachycardia
- Tachycardia
- Ventricular fibrillation
- Urethral burning
- Uterine rupture
References edit
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ a b c d e f "Alprostadil". The American Society of Health-System Pharmacists. Archived from the original on 16 January 2017. Retrieved 8 January 2017.
- ^ Northern Neonatal Network (208). Neonatal Formulary: Drug Use in Pregnancy and the First Year of Life (5 ed.). John Wiley & Sons. p. 2010. ISBN 9780470750353. Archived from the original on 13 January 2017.
- ^ British National Formulary (BNF) (69th ed.). British Medical Association. 2015. p. 569. ISBN 9780857111562.
- ^ Sneader W (2005). Drug Discovery: A History. John Wiley & Sons. p. 185. ISBN 9780470015520. Archived from the original on 13 January 2017.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Meller SM, Stilp E, Walker CN, Mena-Hurtado C (2013). "The link between vasculogenic erectile dysfunction, coronary artery disease, and peripheral artery disease: role of metabolic factors and endovascular therapy". J Invasive Cardiol. 25 (6): 313–9. PMID 23735361.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "Muse Suppository - Facts and Comparisons". Drugs.com. Archived from the original on 19 January 2013. Retrieved 4 January 2013.
- ^ "Edex - Facts and Comparisons". Drugs.com. Archived from the original on 26 October 2012.
- ^ "Caverject - Facts and Comparisons". Drugs.com. Archived from the original on 26 October 2012.
- ^ "Vitaros 3 mg/g cream - Summary of Product Characteristics". Medicines.org.uk. Archived from the original on 11 February 2015.
- ^ "Vitaros- New Erectile Dysfunction Topical Treatment". Meds4All.co.uk. Archived from the original on 11 February 2015.
- ^ Walt RP (November 1992). "Misoprostol for the treatment of peptic ulcer and antiinflammatory-drug-induced gastroduodenal ulceration". The New England Journal of Medicine. 327 (22): 1575–1580. doi:10.1056/NEJM199211263272207. PMID 1435885.
- ^ Wu HL, Marwah S, Wang P, Wang QM, Chen XW (May 2017). "Misoprostol for medical treatment of missed abortion: a systematic review and network meta-analysis". Scientific Reports. 7 (1): 1664. Bibcode:2017NatSR...7.1664W. doi:10.1038/s41598-017-01892-0. PMC 5431938. PMID 28490770.
- ^ Chatsis V, Frey N (2018). Misoprostol for Cervical Ripening and Induction of Labour: A Review of Clinical Effectiveness, Cost-Effectiveness and Guidelines. CADTH Rapid Response Reports. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health. PMID 30907996.
- ^ "Medical abortion". Mayo Clinic. Retrieved 28 April 2022.
- ^ Vietto V, Franco JV, Saenz V, Cytryn D, Chas J, Ciapponi A (January 2018). "Prostanoids for critical limb ischaemia". The Cochrane Database of Systematic Reviews. 1 (1): CD006544. doi:10.1002/14651858.CD006544.pub3. PMC 6491321. PMID 29318581.
- ^ Ye Z, Lu H, Guo W, Dai W, Li H, Yang H, et al. (November 2016). "The effect of alprostadil on preventing contrast-induced nephropathy for percutaneous coronary intervention in diabetic patients: A systematic review and meta-analysis". Medicine. 95 (46): e5306. doi:10.1097/MD.0000000000005306. PMC 5120914. PMID 27861357.
- ^ Xie J, Jiang M, Lin Y, Deng H, Li L (August 2019). "Effect of Alprostadil on the Prevention of Contrast-Induced Nephropathy: A Meta-Analysis of 36 Randomized Controlled Trials". Angiology. 70 (7): 594–612. doi:10.1177/0003319719825597. PMID 30669852. S2CID 58950588.