PMSF: Difference between revisions
m safety advise, difference between inhibitor and inactivator, PMSF inhibits Ser-hydrolases, not only proteases |
Citation bot (talk | contribs) Added bibcode. | Use this bot. Report bugs. | Suggested by Abductive | Category:Serine protease inhibitors | #UCB_Category 2/36 |
||
| (28 intermediate revisions by 17 users not shown) | |||
| Line 2: | Line 2: | ||
| Name = Phenylmethanesulfonyl fluoride (PMSF) |
| Name = Phenylmethanesulfonyl fluoride (PMSF) |
||
| verifiedrevid = 443307656 |
| verifiedrevid = 443307656 |
||
| ImageFile=PMSF.svg |
| ImageFile = PMSF.svg |
||
| ImageSize=180 |
| ImageSize = 180 |
||
| ImageFile1 = PMSF-3D-balls.png |
| ImageFile1 = PMSF-3D-balls.png |
||
| ImageAlt1 = PMSF molecule |
| ImageAlt1 = PMSF molecule |
||
| |
| PIN = Phenylmethanesulfonyl fluoride |
||
| OtherNames= |
| OtherNames = |
||
|Section1={{Chembox Identifiers |
|Section1={{Chembox Identifiers |
||
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
| ChemSpiderID_Ref = {{chemspidercite|correct|chemspider}} |
||
| Line 22: | Line 22: | ||
| StdInChIKey = YBYRMVIVWMBXKQ-UHFFFAOYSA-N |
| StdInChIKey = YBYRMVIVWMBXKQ-UHFFFAOYSA-N |
||
| CASNo_Ref = {{cascite|correct|CAS}} |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| CASNo=329-98-6 |
| CASNo = 329-98-6 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| ⚫ | |||
| UNII = 57KD15003I |
|||
| ⚫ | |||
| ChEBI_Ref = {{ebicite|correct|EBI}} |
| ChEBI_Ref = {{ebicite|correct|EBI}} |
||
| ChEBI = 8102 |
| ChEBI = 8102 |
||
| SMILES = O=S(F)(=O)Cc1ccccc1 |
| SMILES = O=S(F)(=O)Cc1ccccc1 |
||
| MeSHName=Phenylmethylsulfonyl+fluoride |
| MeSHName = Phenylmethylsulfonyl+fluoride |
||
}} |
}} |
||
|Section2={{Chembox Properties |
|Section2={{Chembox Properties |
||
| C=7 | H=7 | F=1 | O=2 | S=1 |
|||
| Formula=C<sub>7</sub>H<sub>7</sub>FO<sub>2</sub>S |
|||
| ⚫ | |||
| MolarMass=174.194 |
|||
| ⚫ | |||
| Density= |
| Density= |
||
| MeltingPt= |
| MeltingPt= |
||
| Line 45: | Line 46: | ||
}} |
}} |
||
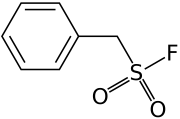
In [[biochemistry]], '''phenylmethylsulfonyl fluoride''' ('''PMSF''') is a [[serine protease |
In [[biochemistry]], '''phenylmethylsulfonyl fluoride''' ('''PMSF''') is a [[serine protease]] inhibitor (serine hydrolase inactivator) commonly used in the preparation of [[cell lysate]]s. PMSF does not inactivate all [[serine protease]]s.<ref>{{Cite journal|url=https://www.jbc.org/article/S0021-9258(19)66050-3/fulltext|doi = 10.1074/jbc.M002758200|doi-access = free|title = Structural Basis for the Insensitivity of a Serine Enzyme (Palmitoyl-Protein Thioesterase) to Phenylmethylsulfonyl Fluoride|year = 2000|last1 = Das|first1 = Amit K.|last2 = Bellizzi|first2 = John J.|last3 = Tandel|first3 = Sagun|last4 = Biehl|first4 = Edward|last5 = Clardy|first5 = Jon|last6 = Hofmann|first6 = Sandra L.|journal = Journal of Biological Chemistry|volume = 275|issue = 31|pages = 23847–23851|pmid = 10801859}}</ref> The effective concentration of PMSF is between 0.1 - 1 mM. The half-life is short in aqueous solutions (110 min at pH 7, 55 min at pH 7.5, and 35 min at pH 8, all at 25 °C).<ref name="James1978">{{cite journal | author = GT James | journal = Analytical Biochemistry | volume = 86 | issue = 2 | pages = 574–9 | year = 1978 | title = Inactivation of the protease inhibitor phenylmethylsulfonyl fluoride in buffers | pmid = 26289 | doi = 10.1016/0003-2697(78)90784-4}}</ref> At 4˚C, pH 8, PMSF is almost completely degraded after 1 day.<ref name="James1978"></ref> Stock solutions are usually made up in anhydrous [[ethanol]], [[isopropanol]], or [[corn oil]] and diluted immediately before use. |
||
PMSF |
PMSF reacts specifically with the active site [[serine]] residue in serine hydrolases. It does not bind to any other serine residues in the protein. This is a result of the hyperactivity of that serine residue caused by the specific environmental conditions in the enzyme's active site ([[catalytic triad]]). Because PMSF bonds covalently to the enzyme, the complex can be viewed by X-ray crystallography; it can therefore be used as a chemical label to identify an essential active site serine in an enzyme. |
||
:Enzyme(active)Ser-O-H + F-SO<sub>2</sub>CH<sub>2</sub>C<sub>6</sub>H<sub>5</sub> → EnzymeSer-O-SO<sub>2</sub>CH<sub>2</sub>C<sub>6</sub>H<sub>5</sub> + HF |
:Enzyme(active)Ser-O-H + F-SO<sub>2</sub>CH<sub>2</sub>C<sub>6</sub>H<sub>5</sub> → EnzymeSer-O-SO<sub>2</sub>CH<sub>2</sub>C<sub>6</sub>H<sub>5</sub> + HF |
||
| Line 53: | Line 54: | ||
:Serine protease + PMSF → Irreversible enzyme-PMS complex + HF |
:Serine protease + PMSF → Irreversible enzyme-PMS complex + HF |
||
The [[median lethal dose]] |
The [[median lethal dose]] between 150–215 mg/kg<ref>{{Cite journal |last1=Myers |first1=D. K. |last2=Kemp |first2=A. |date=January 1954 |title=Inhibition of Esterases by the Fluorides of Organic Acids |url=https://www.nature.com/articles/173033a0 |journal=Nature |language=en |volume=173 |issue=4392 |pages=33–34 |doi=10.1038/173033a0 |pmid=13119739 |bibcode=1954Natur.173...33M |s2cid=4164358 |issn=1476-4687}}</ref><ref>{{Cite journal |last1=Pinsky |first1=C. |last2=Dua |first2=A. K. |last3=LaBella |first3=F. S. |date=Sep 20–27, 1982 |title=Phenylmethylsulfonyl fluoride (PMSF) given systemically produces naloxone-reversible analgesia and potentiates effects of beta-endorphin given centrally |url=https://pubmed.ncbi.nlm.nih.gov/6292607/ |journal=Life Sciences |volume=31 |issue=12–13 |pages=1193–1196 |doi=10.1016/0024-3205(82)90340-x |issn=0024-3205 |pmid=6292607}}</ref> ([[acetylcholine esterase]] inactivator). PMSF should be handled only inside a fume hood and while wearing gloves. [[Dimethyl sulfoxide|DMSO]] is sometimes recommended as solvent for stock solutions, but should not be used as it makes intact skin permeable to PMSF. |
||
==Stability== |
|||
The stability of PMSF in aqueous solutions is low, as it undergoes hydrolysis with water. PMSF is reportedly stable for ~6 months at -20˚C in [[DMSO]],<ref name="Bitesize Bio 2021 m647">{{cite web | title=Protease Inhibitors 101: Best Practices for Use in the Lab | website=Bitesize Bio | date=2021-11-30 | url=https://bitesizebio.com/58195/protease-inhibitors-101/ | access-date=2023-06-22}}</ref> and 9 months at room temperature in 100% [[isopropanol]].<ref name="James1978"></ref><ref>{{cite web |title=The Complete Guide for Protease Inhibition |url=https://iti.stanford.edu/content/dam/sm/iti/documents/himc/immunoassays/ProteaseInhibitionGuide.pdf |publisher=Roche |access-date=22 June 2023}}</ref> |
|||
==Insensitive serine enzymes== |
|||
Some proteins structure limit the accessibility of comparatively bulky PMSF, and therefore PMSF is inactive against these serine enzymes like [[Palmitoyl(protein)_hydrolase|palmitoyl-protein thioesterase]].<ref name="Das2000">{{cite journal | last1=Das | first1=Amit K. | last2=Bellizzi | first2=John J. | last3=Tandel | first3=Sagun | last4=Biehl | first4=Edward | last5=Clardy | first5=Jon | last6=Hofmann | first6=Sandra L. | title=Structural Basis for the Insensitivity of a Serine Enzyme (Palmitoyl-Protein Thioesterase) to Phenylmethylsulfonyl Fluoride | journal=Journal of Biological Chemistry | publisher=Elsevier BV | volume=275 | issue=31 | year=2000 | issn=0021-9258 | doi=10.1074/jbc.m002758200 | pages=23847–23851| pmid=10801859 | doi-access=free }}</ref> Alternative sulfonyl fluoride reagents like [[p-APMSF]] and [[hexadecylsulfonylfluoride|HDSF]], have altered access to native folded protein structures, and may react with serine enzymes that PMSF cannot efficiently react with. This altered selectivity between sulfonyl fluoride reagents has been used to classify and isolate particular types of serine enzymes.<ref name="Das2000"></ref> |
|||
==See also== |
|||
* [[AEBSF]] |
|||
* [[Diisopropyl fluorophosphate|DFP]] (diisopropyl fluorophosphate), an analogous fluorophosphonate phosphorylating reagent |
|||
==References== |
==References== |
||
Latest revision as of 16:41, 26 July 2024
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Phenylmethanesulfonyl fluoride | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.774 |
| KEGG | |
| MeSH | Phenylmethylsulfonyl+fluoride |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C7H7FO2S | |
| Molar mass | 174.19 g·mol−1 |
| Appearance | Powder |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
In biochemistry, phenylmethylsulfonyl fluoride (PMSF) is a serine protease inhibitor (serine hydrolase inactivator) commonly used in the preparation of cell lysates. PMSF does not inactivate all serine proteases.[1] The effective concentration of PMSF is between 0.1 - 1 mM. The half-life is short in aqueous solutions (110 min at pH 7, 55 min at pH 7.5, and 35 min at pH 8, all at 25 °C).[2] At 4˚C, pH 8, PMSF is almost completely degraded after 1 day.[2] Stock solutions are usually made up in anhydrous ethanol, isopropanol, or corn oil and diluted immediately before use.
PMSF reacts specifically with the active site serine residue in serine hydrolases. It does not bind to any other serine residues in the protein. This is a result of the hyperactivity of that serine residue caused by the specific environmental conditions in the enzyme's active site (catalytic triad). Because PMSF bonds covalently to the enzyme, the complex can be viewed by X-ray crystallography; it can therefore be used as a chemical label to identify an essential active site serine in an enzyme.
- Enzyme(active)Ser-O-H + F-SO2CH2C6H5 → EnzymeSer-O-SO2CH2C6H5 + HF
- Serine protease + PMSF → Irreversible enzyme-PMS complex + HF
The median lethal dose between 150–215 mg/kg[3][4] (acetylcholine esterase inactivator). PMSF should be handled only inside a fume hood and while wearing gloves. DMSO is sometimes recommended as solvent for stock solutions, but should not be used as it makes intact skin permeable to PMSF.
Stability
[edit]The stability of PMSF in aqueous solutions is low, as it undergoes hydrolysis with water. PMSF is reportedly stable for ~6 months at -20˚C in DMSO,[5] and 9 months at room temperature in 100% isopropanol.[2][6]
Insensitive serine enzymes
[edit]Some proteins structure limit the accessibility of comparatively bulky PMSF, and therefore PMSF is inactive against these serine enzymes like palmitoyl-protein thioesterase.[7] Alternative sulfonyl fluoride reagents like p-APMSF and HDSF, have altered access to native folded protein structures, and may react with serine enzymes that PMSF cannot efficiently react with. This altered selectivity between sulfonyl fluoride reagents has been used to classify and isolate particular types of serine enzymes.[7]
See also
[edit]References
[edit]- ^ Das, Amit K.; Bellizzi, John J.; Tandel, Sagun; Biehl, Edward; Clardy, Jon; Hofmann, Sandra L. (2000). "Structural Basis for the Insensitivity of a Serine Enzyme (Palmitoyl-Protein Thioesterase) to Phenylmethylsulfonyl Fluoride". Journal of Biological Chemistry. 275 (31): 23847–23851. doi:10.1074/jbc.M002758200. PMID 10801859.
- ^ a b c GT James (1978). "Inactivation of the protease inhibitor phenylmethylsulfonyl fluoride in buffers". Analytical Biochemistry. 86 (2): 574–9. doi:10.1016/0003-2697(78)90784-4. PMID 26289.
- ^ Myers, D. K.; Kemp, A. (January 1954). "Inhibition of Esterases by the Fluorides of Organic Acids". Nature. 173 (4392): 33–34. Bibcode:1954Natur.173...33M. doi:10.1038/173033a0. ISSN 1476-4687. PMID 13119739. S2CID 4164358.
- ^ Pinsky, C.; Dua, A. K.; LaBella, F. S. (Sep 20–27, 1982). "Phenylmethylsulfonyl fluoride (PMSF) given systemically produces naloxone-reversible analgesia and potentiates effects of beta-endorphin given centrally". Life Sciences. 31 (12–13): 1193–1196. doi:10.1016/0024-3205(82)90340-x. ISSN 0024-3205. PMID 6292607.
- ^ "Protease Inhibitors 101: Best Practices for Use in the Lab". Bitesize Bio. 2021-11-30. Retrieved 2023-06-22.
- ^ "The Complete Guide for Protease Inhibition" (PDF). Roche. Retrieved 22 June 2023.
- ^ a b Das, Amit K.; Bellizzi, John J.; Tandel, Sagun; Biehl, Edward; Clardy, Jon; Hofmann, Sandra L. (2000). "Structural Basis for the Insensitivity of a Serine Enzyme (Palmitoyl-Protein Thioesterase) to Phenylmethylsulfonyl Fluoride". Journal of Biological Chemistry. 275 (31). Elsevier BV: 23847–23851. doi:10.1074/jbc.m002758200. ISSN 0021-9258. PMID 10801859.
External links
[edit]- The MEROPS online database for peptidases and their inhibitors: PMSF
