Gut microbiota: Difference between revisions
ClueBot NG (talk | contribs) m Reverting possible vandalism by 39.32.158.54 to version by Jytdog. Report False Positive? Thanks, ClueBot NG. (2597105) (Bot) |
|||
| Line 136: | Line 136: | ||
Weight loss initiates a shift in the bacteria phyla that compose gut flora. Specifically, ''Bacteroidetes'' increase nearly linearly as weight loss progresses.<ref name="Ley2006">{{cite journal |doi=10.1038/4441022a |title=Microbial ecology: Human gut microbes associated with obesity |year=2006 |last1=Ley |first1=Ruth E. |last2=Turnbaugh |first2=Peter J. |last3=Klein |first3=Samuel |last4=Gordon |first4=Jeffrey I. |journal=Nature |volume=444 |issue=7122 |pages=1022–3 |pmid=17183309}}</ref> While there is a high level of variation in bacteria species found among individual people, this trend is prominent and distinct in humans.<ref>{{cite journal |doi=10.1111/j.1574-6941.2002.tb00904.x |title=Assessment of microbial diversity in human colonic samples by 16S rDNA sequence analysis |year=2002 |last1=Hold |first1=Georgina L. |last2=Pryde |first2=Susan E. |last3=Russell |first3=Valerie J. |last4=Furrie |first4=Elizabeth |last5=Flint |first5=Harry J. |journal=FEMS Microbiology Ecology |volume=39 |pages=33–9 |pmid=19709182 |issue=1}}</ref> |
Weight loss initiates a shift in the bacteria phyla that compose gut flora. Specifically, ''Bacteroidetes'' increase nearly linearly as weight loss progresses.<ref name="Ley2006">{{cite journal |doi=10.1038/4441022a |title=Microbial ecology: Human gut microbes associated with obesity |year=2006 |last1=Ley |first1=Ruth E. |last2=Turnbaugh |first2=Peter J. |last3=Klein |first3=Samuel |last4=Gordon |first4=Jeffrey I. |journal=Nature |volume=444 |issue=7122 |pages=1022–3 |pmid=17183309}}</ref> While there is a high level of variation in bacteria species found among individual people, this trend is prominent and distinct in humans.<ref>{{cite journal |doi=10.1111/j.1574-6941.2002.tb00904.x |title=Assessment of microbial diversity in human colonic samples by 16S rDNA sequence analysis |year=2002 |last1=Hold |first1=Georgina L. |last2=Pryde |first2=Susan E. |last3=Russell |first3=Valerie J. |last4=Furrie |first4=Elizabeth |last5=Flint |first5=Harry J. |journal=FEMS Microbiology Ecology |volume=39 |pages=33–9 |pmid=19709182 |issue=1}}</ref> |
||
=== '''Splenda''' === |
|||
| ⚫ | |||
Recent studies have shown the effects of Splenda in altering gut flora. Each year, millions of people use Splenda as an alternative to sugar and other sweeteners.<ref>{{Cite web|url=http://www.cnn.com/2016/01/18/health/where-do-we-stand-artificial-sweeteners/index.html|title=Health effects of artificial sweeteners|website=CNN|access-date=2016-03-24}}</ref> If this research is found to be valid, millions of people could be altering their gut bacteria, and thus affecting their metabolism, mood, weight, nutrient absorption, and immune system.<ref>{{Cite web|url=http://www.cnn.com/2014/06/18/health/good-gut-bacteria/index.html|title=Bacteria in your gut: 4 healthy habits - CNN.com|website=CNN|access-date=2016-03-24}}</ref> |
|||
| ⚫ | Bacteria in the digestive tract have pathogenic properties in addition to their health-inducing ones: they can produce [[toxin]]s and [[carcinogen]]s{{citation needed|date=October 2013}} and have been implicated in such conditions as [[multisystem organ failure]], [[sepsis]], [[colon cancer]], and [[inflammatory bowel disease]] (IBD).<ref name="Guarner and Malagelada 2003b"/> A major factor in health is the balance of bacterial numbers; if the numbers grow too high or low, it will result in harm to the host. The host has [[enzyme]]s to regulate this balance.{{citation needed|date=October 2013}} |
||
At the end a12 week study, the numbers of total anaerobes, bifidobacteria, lactobacilli, Bacteroides, clostridia, and total aerobic bacteria were significantly decreased in the test subjects.<ref>{{Cite journal|last=Abou-Donia|first=Mohamed B.|last2=El-Masry|first2=Eman M.|last3=Abdel-Rahman|first3=Ali A.|last4=McLendon|first4=Roger E.|last5=Schiffman|first5=Susan S.|date=2008-01-01|title=Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome p-450 in male rats|url=http://www.ncbi.nlm.nih.gov/pubmed/18800291|journal=Journal of Toxicology and Environmental Health. Part A|volume=71|issue=21|pages=1415–1429|doi=10.1080/15287390802328630|issn=1528-7394|pmid=18800291}}</ref> Conclusions of the study stated that Splenda’s adverse side effects included a reduction in beneficial gut microflora. |
|||
Controversy has arisen surrounding the validity of the claims of such studies. The test subjects used were rats, which are biologically different than human beings. Furthermore, the dose per kilogram of body weight also could severely alter the results of the study. Another study found that the bacterial consortia cannot fully digest Splenda, so a dose of about 300 ng/L should not have any effect on bacterial growth.<ref>{{Cite web|url=http://www.hindawi.com/journals/jt/2013/372986/#B7|title=Metabolic Effects of Sucralose on Environmental Bacteria|last=Omran|first=Arthur|date=10 November 2013|website=Hindawi Publishing Corporation|publisher=Journal of Toxicology|access-date=21 March 2016}}</ref> |
|||
Findings seem to show that the ratio of Splenda to gut liquid volume influences the effect of Splenda upon the gut micro flora. At high doses, reduction of bacteria can be observed. However, at low doses relative to gut volume, Splenda does not seem to have significant impacts upon gut bacterium growth. |
|||
| ⚫ | |||
| ⚫ | Bacteria in the digestive tract have pathogenic properties in addition to their health-inducing ones: they can produce [[toxin]]s and [[carcinogen]]s{{citation needed|date=October 2013}} and have been implicated in such conditions as [[multisystem organ failure]], [[sepsis]], [[colon cancer]], and [[inflammatory bowel disease]] (IBD).<ref name="Guarner and Malagelada 2003b" /> A major factor in health is the balance of bacterial numbers; if the numbers grow too high or low, it will result in harm to the host. The host has [[enzyme]]s to regulate this balance.{{citation needed|date=October 2013}} |
||
===Cancer=== |
===Cancer=== |
||
Revision as of 15:11, 24 March 2016
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
|

Gut flora or, more appropriately, gut microbiota, consists of a complex community of microorganisms that live in the digestive tracts of animals. The gut microbiota comprises the largest and most diverse reservoir of mutualistic microorganisms associated with animals. In this context gut is synonymous with intestinal, and flora with microbiota and microflora. An associated term, sometimes used interchangeably with gut microbiota, is gut microbiome, which refers to the aggregate of all the genomes of gut microbiota.
Gut microorganisms benefit the host by collecting the energy from the fermentation of undigested carbohydrates and the subsequent absorption of short-chain fatty acids. The most important of these fatty acids are butyrates, metabolised by the colonic epithelium; propionates by the liver; and acetates by the muscle tissue. Intestinal bacteria also play a role in synthesizing vitamin B and vitamin K as well as metabolizing bile acids, sterols and xenobiotics.[1][2][3][4][5][6] The metabolic activities performed by these bacteria resemble those of an organ, leading some to liken gut bacteria to a "forgotten" organ. It is estimated that these gut flora have around a hundred times as many genes in aggregate as there are in the human genome.[7]
Different people and animals have different types of microorganisms in their guts. The composition of the gut flora also changes over time, when the diet changes.
Description
The majority of the microbial content of the colon consists of bacteria. They also make up to 60% of the dry mass of feces.[8] Somewhere between 300[3] and 1000 different species live in the gut,[4] with most estimates at about 500.[5][9][10] However, it is probable that 99% of the bacteria come from about 30 or 40 species.[11] Fungi, protozoa, and archaea also make up a part of the gut flora, but little is known about their activities.
Research suggests that the relationship between gut flora and humans is not merely commensal (a non-harmful coexistence), but rather a mutualistic relationship.[4] Though people can survive without gut flora,[5] the microorganisms perform a host of useful functions, such as fermenting unused energy substrates, training the immune system, preventing growth of harmful, pathogenic bacteria,[3] regulating the development of the gut, producing vitamins for the host, such as biotin[12] and vitamin K,[13] and producing hormones to direct the host to store fats. In return, these microorganisms procure within the host a protected, nutrient-rich environment in which they can thrive. However, in certain conditions, some species are thought to be capable of causing disease by producing infection or increasing cancer risk for the host.[3]
Over 99% of the bacteria in the gut are anaerobes,[3][4][11][14] but in the cecum, aerobic bacteria reach high densities.[3]
The compositions of microbiota rely on several factors like host diet, colonization history, and immune status.[15] Some microbes are better suited to complement specific metabolic enzymes over others. For example, Bacteroides change how they break down carbohydrates depending on if they received the right amount of certain substrates.[16]
Aside from mammals, some insects also possess complex and diverse gut microbiota that play key nutritional roles.[17] Microbial communities associated termites can constitute a majority of the weight of the individuals and perform important roles in the digestion of lignocellulose and nitrogen fixation.[18] These communities are host-specific, and closely related insect species share comparable similarities in gut microbiota composition.[19][20] In cockroaches, gut microbiota have been shown to assemble in a deterministic fashion, irrespective of the inoculum;[21] the reason for this host-specific assembly remains unclear. Bacterial communities associated with insects like termites and cockroaches are determined by a combination of forces, primarily diet, but there is some indication that host phylogeny may also be playing a role in the selection of lineages.[19][20]
Types
Not all the species in the gut have been identified[3][4] because most cannot be cultured,[4][11][22] and identification is difficult. Populations of species vary widely among different individuals but stay fairly constant within an individual over time, even though some alterations may occur with changes in lifestyle, diet and age.[3][9] An effort to better describe the microflora of the gut and other body locations has been initiated; see Human microbiome project. In 2009, scientists from INRA (France) highlighted the existence of a small number of species shared by all individuals constituting the human intestinal microbiota phylogenetic core.[23]
The four dominant phyla in the human gut are Firmicutes, Bacteroidetes, Actinobacteria, and Proteobacteria.[24] Most bacteria belong to the genera Bacteroides, Clostridium, Faecalibacterium,[3][11][14] Eubacterium, Ruminococcus, Peptococcus, Peptostreptococcus, and Bifidobacterium.[3][11] Other genera, such as Escherichia and Lactobacillus, are present to a lesser extent.[3] Species from the genus Bacteroides alone constitute about 30% of all bacteria in the gut, suggesting that this genus is especially important in the functioning of the host.[4]
The currently known genera of fungi of the gut flora include Candida, Saccharomyces, Aspergillus, and Penicillium.
Archaea constitute another large class of gut flora which are important in the metabolism of the bacterial products of fermentation.
Enterotype
An enterotype is a classification of living organisms based on its bacteriological ecosystem in the human gut microbiome not dictated by age, gender, body weight, or national divisions.[25] There are indications that long-term diet influences enterotype.[26] Three human enterotypes have been discovered.[25][27]
Age
It has been demonstrated that there are common patterns of microbiome composition evolution during life.[28] Analysing bacterial V4 16S rRNA from 528 individuals of different ages and geographic origins,[29] demonstrated that the diversity of microbiota composition of the fecal samples is significantly higher in adults than in children, although interpersonal differences are higher in children than in adults. Interestingly, the maturation of microbiota into an adult-like configuration happens during the three first years of life. The metagenomic analysis of fecal samples combined with the analysis of V4 16S rRNA allowed a detailed study of phylotypes and showed that although there are not phylotypes being unique to adults or babies, phylotypes belonging to Bifidobacteria longum which dominate breast fed babies, decline in proportional representation with increasing age.[29]
The study also showed a high prevalence of enzymes involved in fermentation, methanogenesis and in the metabolism of arginine, glutamate, aspartate and lysine in adult microbiomes whereas in infant microbiomes the dominant enzymes are involved in cysteine metabolism and fermentation pathways.[29]
Finally, analysing the effects of kinship on the microbiome across countries, it has been found that despite the large influence of cultural factors on which microbes are present in the population, sharing numerous common environmental exposures in a family is a strong determinant of individual microbiome composition. This effect has no genetic influence and it is consistently observed in culturally different populations.[29]
Gut flora and diet
Studies and statistical analyses have identified the different bacterial genera in gut microbiota and their associations with nutrient intake. Gut microflora is mainly composed of three enterotypes: Prevotella, Bacteroides and Ruminococcus. There is an association between the concentration of each microbial community and dietary components. For example, Prevotella is related to carbohydrates and simple sugars, indicating an association with a carbohydrate-based diet more typical of agrarian societies, while Bacteroides is associated with animal proteins, amino acids and saturated fats, components typical of a Western diet. That means that one enterotype will dominate over the other depending on the diet - those who eat plenty of protein and animal fats have predominantly Bacteroides bacteria, while for those who consume more carbohydrates the Prevotella species dominate.[26]
Gut microbiome however can be changed by following a long-term diet. People whose microbiome is predominantly Bacteroides (diet based on high levels of protein and fat) and change their dietary patterns to a diet based on high levels of carbohydrates, will develop a Prevotella enterotype in the long-term. This relation may be interesting medically, as long-term dietary interventions may allow modulation of an individual's enterotype to improve health.[26]
Geography
Gut microbiome composition depends on the geographic origin of populations. Variations in trade off of Prevotella, the representation of the urease gene, and the representation of genes encoding glutamate synthase/degradation or other enzymes involved in amino acids degradation or vitamin biosynthesis show significant differences between populations from USA, Malawi or Amerindian origin.[29]
The US population has a high representation of enzymes encoding the degradation of glutamine and enzymes involved in vitamin and lipoic acid biosynthesis; whereas Malawi and Amerindian populations have a high representation of enzymes encoding glutamate synthase and they also have an overrepresentation of
Further studies have indicated a large difference in the composition of microbiota between European and rural African children. The fecal bacteria of children from Florence were compared to that of children from the small rural village of Boulpon in Burkina Faso. The diet of a typical child living in this village is largely lacking in fats and animal proteins and rich in polysaccharides and plant proteins. The fecal bacteria of European children was dominated by Firmicutes and showed a marked reduction in biodiversity, while the fecal bacteria of the Boulpon children was dominated by Bacteroidetes. The increased biodiversity and different composition of gut flora in African populations may aid in the digestion of normally indigestible plant polysaccharides and also may result in a reduced incidence of non-infectious colonic diseases.[30]
Acquisition of gut flora in human infants
The gastrointestinal tract of a normal fetus has been considered to be sterile, however recently it has been acknowledged that microbial colonisation may occur in the fetus.[31] During birth and rapidly thereafter, bacteria from the mother and the surrounding environment colonize the infant's gut. Immediately after vaginal delivery, babies may have bacterial strains derived from the mothers' feces in the upper gastrointestinal tract.[32] Infants born by caesarean section may also be exposed to their mothers' microflora, but the initial exposure is most likely to be from the surrounding environment such as the air, other infants, and the nursing staff, which serve as vectors for transfer.[33] The initial inoculum of babies born by cesarian section is typically dominated by Staphyloccocus, Corynebacterium and Propionibacterium species.[34] The primary gut flora in infants born by caesarean delivery may be disturbed for up to six months after birth, whereas vaginally born infants take up to one month for their intestinal microflora to be well established.[35] After birth, environmental, oral and cutaneous bacteria are readily transferred from the mother to the infant through suckling, kissing, and caressing.
All infants are initially colonized by large numbers of E. coli and streptococci. Within a few days, bacterial numbers reach 108 to 1010 per gram of feces.[33][36] During the first week of life, these bacteria create a reducing environment favorable for the subsequent bacterial succession of strict anaerobic species mainly belonging to the genera Bifidobacterium, Bacteroides, Clostridium, and Ruminococcus.[37] Breast-fed babies become dominated by bifidobacteria, possibly due to the contents of bifidobacterial growth factors in breast milk.[38][39] In contrast, the microbiota of formula-fed infants is more diverse, with high numbers of Enterobacteriaceae, enterococci, bifidobacteria, Bacteroides, and clostridia.[40][41]
The microbiomes of infants are enriched by enzymes involved in foraging of glycans represented in breast milk and the intestinal mucosa.[29]
Functions
Bacteria in the gut fulfill a host of useful functions for humans, including digestion of unutilized energy substrates,[42] stimulating cell growth, repressing the growth of harmful microorganisms, training the immune system to respond only to pathogens, and defending against some diseases.[3][4][43]
Carbohydrate fermentation and absorption
Without gut flora, the human body would be unable to utilize some of the undigested carbohydrates it consumes, because some types of gut flora have enzymes that human cells lack for breaking down certain polysaccharides.[4] Rodents raised in a sterile environment and lacking in gut flora need to eat 30% more calories just to remain the same weight as their normal counterparts.[4] Carbohydrates that humans cannot digest without bacterial help include certain starches, fiber, oligosaccharides and sugars that the body failed to digest and absorb[3][10][11] like lactose in the case of lactose intolerance and sugar alcohols, mucus produced by the gut, and proteins.[10] A further result is flatulence, specifically due to the metabolism of oligosaccharides (notably from beans) by many different species.
Bacteria turn carbohydrates they ferment into short-chain fatty acids (SCFAs)[10][11] by a form of fermentation called saccharolytic fermentation.[10] Products include acetic acid, propionic acid and butyric acid.[10][11] These materials can be used by host cells, providing a major source of useful energy and nutrients for humans,[10] as well as helping the body to absorb essential dietary minerals such as calcium, magnesium and iron.[3] Gases and organic acids, such as lactic acid, are also produced by saccharolytic fermentation.[11] Acetic acid is used by muscle, propionic acid helps the liver produce ATP, and butyric acid provides energy to gut cells and may prevent cancer.[10] Evidence also indicates that bacteria enhance the absorption and storage of lipids[4] and produce and then facilitate the body to absorb needed vitamins like vitamin K.
Beneficial flora increase the gut's absorption of water, reduce counts of damaging bacteria, increase growth of human gut cells,[citation needed] and stimulate growth of indigenous bacteria.[3]
Trophic effects
Another benefit of SCFAs is that they increase growth of intestinal epithelial cells and control their proliferation and differentiation.[3] They may also cause lymphoid tissue near the gut to grow. Bacterial cells also alter intestinal growth by changing the expression of cell surface proteins such as sodium/glucose transporters.[4] In addition, changes they make to cells may prevent injury to the gut mucosa from occurring.[43]
Suppression of pathogenic microbial growth
Another important role of helpful gut flora is that they prevent species that would harm the host from colonizing the gut through competitive exclusion, an activity termed the "barrier effect". Harmful yeasts and bacterial species such as Clostridium difficile (the overgrowth of which can cause pseudomembranous colitis) are unable to grow excessively due to competition from helpful gut flora species adhering to the mucosal lining of the intestine, thus animals without gut flora are infected very easily. The barrier effect protects humans from both invading species and species normally present in the gut at low numbers, whose growth is usually inhibited by the gut flora.[3]
Helpful bacteria prevent the growth of pathogenic species by competing for nutrition and attachment sites to the epithelium of the colon. Symbiotic bacteria are more at home in this ecological niche and are thus more successful in the competition. Indigenous gut floras also produce bacteriocins, which are proteinaceous toxins that inhibit growth of similar bacterial strains, substances that kill harmful microbes and the levels of which can be regulated by enzymes produced by the host.[3]
The process of fermentation, since it produces lactic acid and different fatty acids, also serves to lower the pH in the colon, preventing the proliferation of harmful species of bacteria and facilitating that of helpful species. The pH may also enhance the excretion of carcinogens.[10]
Immunity
This section's factual accuracy is disputed. (October 2015) |
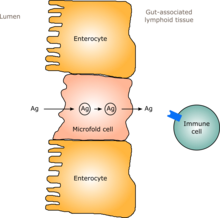
Gut flora affects the systemic immune systems by promoting the early development of the gut's mucosal immune system respective to its physical components and function. Subsequently the bacteria perpetuate the impact on the immune system functions. The intestinal microbiota stimulates the lymphoid tissue associated with the gut mucosa to produce antibodies to pathogens. Thus the immune system learns to recognize and fight harmful bacteria and develops tolerance for useful species.[3][5][22]
After childbirth, bacteria begin colonizing the body including the digestive tract. This first colonization of the gut forms the person's lifelong gut flora. The first microbes modulate the immune response, since bacteria attempt to make it more favorable to their own survival and less to competing species. However, there is a shift at the time of weaning from predominantly facultative anaerobic species, such as Streptococci and Escherichia coli, to mostly obligate anaerobic species.[3][4]
Bacteria can influence the phenomenon known as oral tolerance, in which the immune system is less sensitive to an antigen (including those produced by gut bacteria) once it has been ingested. Partly the gastrointestinal immune system and partly the liver mediate this tolerance, which can reduce an overreactive immune response like those found in allergies and autoimmune disease.[44] Oral tolerance is dependent on the maturation of regulatory T cells (Treg) that evolve in response to signals from the gut flora delivered by other immune cells. Consequently different species of gut microbiota have different impact on the cells of the immune system and the probability to acquire oral tolerance.[45] The ability of the gut flora to exert these immunomodulatory effects on the host appears to be restricted to a ‘critical window’ during development when the flora population is first becoming established and the immune system is completing its ripening.[46] Latest research results support this hypothesis by disclosing associations between altered patterns of microbial colonization during this critical window and an increased likelihood of developing atopic diseases.[47]
Recent findings revealed that gut microbiota metabolism of dietary fiber influences allergic airway disease. This is induced by the metabolism of the fiber via the gut microbiota and thereby increased local and systemic levels of short-chain fatty acids, which leads to changes in the composition of the lung microbiota. An increased amount of circulating short-chain fatty acids influences dendritic cell hematopoiesis and functionality. The effects of short-chain fatty acids on allergic inflammation are dependent on G protein-coupled receptor 41.[48]
Once the host immune system is developed, it regulates the bacterial composition in the gut. One of the regulation mechanisms—immune exclusion—is mediated through the neutralizing action of secretory immunoglobulin A (IgA).[49]
Metabolic function
The resident gut microflora positively control the intestinal epithelial cell differentiation and proliferation through the production of short-chain fatty acids. They also mediate other metabolic effects such as the syntheses of vitamins like biotin and folate, as well as absorption of ions including magnesium, calcium and iron.[9] Methanogenic archae such as Methanobrevibacter smithii are involved in the removal of end products of bacterial fermentation such as hydrogen.
Preventing allergy
Bacteria are implicated in preventing allergies,[2] which are overreactions of the immune system to harmless antigens. Studies on the gut flora of infants and young children have shown that those who have or later develop allergies have different compositions of gut flora from those without allergies, with higher chances of having the harmful species C. difficile and S. aureus and lower prevalence of Bacteroides and Bifidobacteria.[2] One explanation is that since helpful gut flora stimulate the immune system and "train" it to respond properly to antigens, a lack of these bacteria in early life leads to an inadequately trained immune system that overreacts to antigens.[2] On the other hand, the differences in flora could be a result, not a cause, of the allergies.[2]
Preventing inflammatory bowel disease
Another indicator that bacteria help train the immune system is the epidemiology of inflammatory bowel disease, or IBD, such as Crohn's disease (CD). Some authors suggest that SCFAs prevent IBD. In addition, some forms of bacteria can prevent inflammation.[50] The incidence and prevalence of IBD is high in industrialized countries with a high standard of living and low in less economically developed countries, having increased in developed countries throughout the twentieth century. The disease is also linked to good hygiene in youth; lack of breastfeeding; and consumption of large amounts of sucrose and animal fat.[50] Its incidence is inversely linked with poor sanitation during the first years of life and consumption of fruits, vegetables, and unprocessed foods.[50] Also, the use of antibiotics, which kill native gut flora and harmful infectious pathogens alike, especially during childhood, is associated with inflammatory bowel disease.[42] On the other hand, using high concentrations of probiotics called "Amanlac" probiotics, helps to effectively maintain intestinal homeostasis, while also treating intestinal inflammation in IBD.[51]
Alterations in flora balance
Inflammation
Many tests with colitis-susceptible mice (Il10−/−) show significantly increased levels of luminal Verrucomicrobia, Bacteroidetes and Proteobacteria. For example, inflammation promotes an E. coli proliferation.
Effects of antibiotic use
Altering the numbers of gut bacteria, for example by taking broad-spectrum antibiotics, may affect the host's health and ability to digest food.[52] Antibiotics can cause antibiotic-associated diarrhea (AAD) by irritating the bowel directly, changing the levels of gut flora, or allowing pathogenic bacteria to grow.[11] Another harmful effect of antibiotics is the increase in numbers of antibiotic-resistant bacteria found after their use, which, when they invade the host, cause illnesses that are difficult to treat with antibiotics.[52]
Changing the numbers and species of gut flora can reduce the body's ability to ferment carbohydrates and metabolize bile acids and may cause diarrhea. Carbohydrates that are not broken down may absorb too much water and cause runny stools, or lack of SCFAs produced by gut flora could cause the diarrhea.[11]
A reduction in levels of native bacterial species also disrupts their ability to inhibit the growth of harmful species such as C. difficile and Salmonella kedougou, and these species can get out of hand, though their overgrowth may be incidental and not be the true cause of diarrhea.[3][11][52] Emerging treatment protocols for C. difficile infections involve fecal microbiota transplantation of donor feces. (see Fecal transplant). Initial reports of treatment describe success rates of 90%, with few side effects. Efficacy is speculated to result from restoring bacterial balances of bacteroides and firmicutes classes of bacteria.[53]
Gut flora composition also changes in severe illnesses, due not only to antibiotic use but also to such factors as ischemia of the gut, failure to eat, and immune compromise. Negative effects from this have led to interest in selective digestive tract decontamination (SDD), a treatment to kill only pathogenic bacteria and allow the re-establishment of healthy ones.[54]
For more than 51 years we have known that the administration of low doses of antibacterial agents promotes the growth of farm animals to increase weight gain.[55]
Antibiotics alter the population of the gastrointestinal (GI) tract microbiota, may change the intra-community metabolic interactions, modify caloric intake by using carbohydrates, and globally affects host metabolic, hormonal and immune homeostasis.[55]
In a study performed on mice by Ilseung Cho,[55] the ratio of Firmicutes and Lachnospiraceae was significantly elevated in animals treated with subtherapeutic doses of different antibiotics. By analyzing the caloric content of faeces and the concentration of small chain fatty acids (SCFAs) in the GI tract, they concluded that the changes in the composition of microbiota lead to an increased capacity to extract calories from otherwise indigestible constituents, and to an increased production of SCFAs. These findings provide evidence that antibiotics perturb not only the composition of the GI microbiome but also its metabolic capabilities, specifically with respect to SCFAs.[55]
Pharmabiotics
Pharmabiotics is a generic term to encompass any form of therapeutic exploitation of the commensal flora, including the use of live probiotic bacteria, probiotic-derived biologically active metabolites, prebiotics, synbiotics or genetically modified commensal bacteria.[9] Since the lack of gut flora can have such harmful health effects, the use of probiotics has anti-inflammatory effects in the gut and may be useful for improving health. Prebiotics are dietary components that can help foster the growth of micro-organisms in the gut, which may lead to better health.[50] There is evidence supporting a therapeutic role for probiotic strategies for treating mucosal inflammatory disorders such as IBD, atopy, infection, diarrhoea, cancer and arthritis.[9]
Pregnancy
Women's gut microbiota change as pregnancy advances, with the changes similar to those seen in metabolic syndromes such as diabetes. The change in gut flora causes no ill effects. The newborn's gut biota resemble the mother's first-trimester samples. The diversity of the flora decreases from the first to third trimester, as the numbers of certain species go up.[56]
Weight loss
Weight loss initiates a shift in the bacteria phyla that compose gut flora. Specifically, Bacteroidetes increase nearly linearly as weight loss progresses.[57] While there is a high level of variation in bacteria species found among individual people, this trend is prominent and distinct in humans.[58]
Splenda
Recent studies have shown the effects of Splenda in altering gut flora. Each year, millions of people use Splenda as an alternative to sugar and other sweeteners.[59] If this research is found to be valid, millions of people could be altering their gut bacteria, and thus affecting their metabolism, mood, weight, nutrient absorption, and immune system.[60]
At the end a12 week study, the numbers of total anaerobes, bifidobacteria, lactobacilli, Bacteroides, clostridia, and total aerobic bacteria were significantly decreased in the test subjects.[61] Conclusions of the study stated that Splenda’s adverse side effects included a reduction in beneficial gut microflora.
Controversy has arisen surrounding the validity of the claims of such studies. The test subjects used were rats, which are biologically different than human beings. Furthermore, the dose per kilogram of body weight also could severely alter the results of the study. Another study found that the bacterial consortia cannot fully digest Splenda, so a dose of about 300 ng/L should not have any effect on bacterial growth.[62]
Findings seem to show that the ratio of Splenda to gut liquid volume influences the effect of Splenda upon the gut micro flora. At high doses, reduction of bacteria can be observed. However, at low doses relative to gut volume, Splenda does not seem to have significant impacts upon gut bacterium growth.
Role in disease
Bacteria in the digestive tract have pathogenic properties in addition to their health-inducing ones: they can produce toxins and carcinogens[citation needed] and have been implicated in such conditions as multisystem organ failure, sepsis, colon cancer, and inflammatory bowel disease (IBD).[3] A major factor in health is the balance of bacterial numbers; if the numbers grow too high or low, it will result in harm to the host. The host has enzymes to regulate this balance.[citation needed]
Cancer
Some genera of bacteria, such as Bacteroides and Clostridium, have been associated with an increase in tumor growth rate, while other genera, such as Lactobacillus and Bifidobacteria, are known to prevent tumor formation.[3]
Translocation
Helpful bacteria can be very harmful to the host if they get outside of the intestinal tract.[4][14] Translocation, which occurs when bacteria leave the gut through its mucosal lining, the border between the lumen of the gut and the inside of the body,[5][63] can occur in a number of different diseases.[14][50] It can be caused by too much growth of bacteria in the small intestine, reduced immunity of the host, or increased gut lining permeability.[50] The gut can become more permeable in diseases like cirrhosis, which is damaging due in part to the activity of gut flora.[64]
If the gut is perforated, bacteria can invade the body, causing a potentially fatal infection. Aerobic bacteria can make an infection worse by using up all available oxygen and creating an environment favorable to anaerobes.[14]
Likewise, just recently it was discovered that stomach ulcers could be caused by Helicobacter pylori sneaking underneath the epithelial lining of the stomach. Here the body produces an immune response. During this response parietal cells are stimulated and release extra hydrochloric acid (HCl+) into the stomach. However, the response does not stimulate the mucus-secreting cells that protect and line the epithelium of the stomach. The extra acid translates to searing holes into the epithelial lining of the stomach, resulting in stomach ulcers. Moreover, negatively affected microbiota compositions have a detrimental affect on individuals. Research experiments show that one of the probable causes for inflammatory bowel disorder (chronic inflammatory disorders – Crohn's disease), Likewise, an imbalance in microbiota levels can cause widespread diseases, such as irritable bowel syndrome (functional bowel disorder) and obesity.[28]
Inflammatory bowel disease
Some suspect that IBD is due to a reduction in immune tolerance and subsequent overreaction of the host's immune system to harmful or non-harmful bacteria. IBD may be caused by the entire gut flora together or some specific types.[42][65]
It has been noted that though ulcerative colitis and Crohn's disease (two types of IBD) probably have genetic components, they are not inherited in a Mendelian fashion and are thus probably due to a complex set of factors rather than solely to a gene.[65] Though neither bacterial colonization nor genetics is sufficient to cause the disease, bacteria probably play a role in these disorders.[65]
Some suspect that inflammation in IBD is due to increased permeability of the inner lining of the colon, which may allow bacteria to invade the tissues and cause an immune reaction that leads to prolonged inflammation.[5][63] Tissue damage in IBD results from the immunological misperception of danger within the naturally occurring flora or due to failure of normal tolerance to pathogenic bacteria. It is still unclear whether the inflammation that occurs is due to a specific subset of intestinal microbes or due to a problem with the tolerance of commensal gut flora.[9] Abnormal tight junctions, which are supposed to prevent permeability, have been found in cells of patients with IBD.[63] Because of the potentially harmful role of these bacteria, antibiotics are frequently prescribed to treat Crohn's disease.[43] However, inflammation could occur first and cause the increased intestinal permeability found in diseases such as Crohn's, so the causative role of bacteria is not clear.[63] Conventional therapies for IBD primarily target the mucosal inflammatory responses by using pharmabiotics.[9]
Colitis
It has been suggested that commensal bacteria are responsible for the development of colitis, since mice raised in a sterile environment do not get the disease.[66] However, while some bacterial strains such as C. difficile[50] and even normal gut bacteria cause colitis,[66] others prevent the disease in mice.[50]
Obesity
The mutual influence of gut flora composition and weight condition is connected to differences in the energy-reabsorbing potential of different ratios of Firmicutes and Bacteroidetes, especially in the digestion of fatty acids and dietary polysaccharides, as shown by experiments[67] wherein the (caecum) gut flora of obese mice were transplanted into germ-free recipient mice, leading to an increase in weight despite a decrease in food consumption. The experimentation and results suggest bacteria specific to lean and obese genes can be a weapon in the fight against the obesity epidemic.[57][68][69][70][71]
See also
- Colonisation resistance
- Helminthic therapy
- Human microbiome
- Human microbiome project
- List of human flora
- Microbiome
- Probiotic
- Segmented filamentous bacteria
- Skin flora
- uBiome
- Verotoxin-producing Escherichia coli
- Fecal bacteriotherapy
- List of microbiota species of the lower reproductive tract of women
Sources and notes
- ^ Cummings, J.H.; MacFarlane, G.T. (1997). "Role of intestinal bacteria in nutrient metabolism". Clinical Nutrition. 16: 3–9. doi:10.1016/S0261-5614(97)80252-X.
- ^ a b c d e Björkstén, Bengt; Sepp, Epp; Julge, Kaja; Voor, Tiia; Mikelsaar, Marika (2001). "Allergy development and the intestinal microflora during the first year of life". Journal of Allergy and Clinical Immunology. 108 (4): 516–20. doi:10.1067/mai.2001.118130. PMID 11590374.
- ^ a b c d e f g h i j k l m n o p q r s t u v w Guarner, F; Malagelada, J (2003). "Gut flora in health and disease". The Lancet. 361 (9356): 512–9. doi:10.1016/S0140-6736(03)12489-0. PMID 12583961.
- ^ a b c d e f g h i j k l m n Sears, Cynthia L. (2005). "A dynamic partnership: Celebrating our gut flora". Anaerobe. 11 (5): 247–51. doi:10.1016/j.anaerobe.2005.05.001. PMID 16701579.
- ^ a b c d e f Steinhoff, U (2005). "Who controls the crowd? New findings and old questions about the intestinal microflora". Immunology Letters. 99 (1): 12–6. doi:10.1016/j.imlet.2004.12.013. PMID 15894105.
- ^ Savage, D C (1977). "Microbial Ecology of the Gastrointestinal Tract". Annual Review of Microbiology. 31: 107–33. doi:10.1146/annurev.mi.31.100177.000543. PMID 334036.
- ^ Qin, Junjie; Li, Ruiqiang; Raes, Jeroen; Arumugam, Manimozhiyan; Burgdorf, Kristoffer Solvsten; Manichanh, Chaysavanh; Nielsen, Trine; Pons, Nicolas; Levenez, Florence; Yamada, Takuji; Mende, Daniel R.; Li, Junhua; Xu, Junming; Li, Shaochuan; Li, Dongfang; Cao, Jianjun; Wang, Bo; Liang, Huiqing; Zheng, Huisong; Xie, Yinlong; Tap, Julien; Lepage, Patricia; Bertalan, Marcelo; Batto, Jean-Michel; Hansen, Torben; Le Paslier, Denis; Linneberg, Allan; Nielsen, H. Bjørn; Pelletier, Eric; Renault, Pierre (2010). "A human gut microbial gene catalogue established by metagenomic sequencing". Nature. 464 (7285): 59–65. doi:10.1038/nature08821. PMC 3779803. PMID 20203603.
- ^ Stephen, A. M.; Cummings, J. H. (1980). "The Microbial Contribution to Human Faecal Mass". Journal of Medical Microbiology. 13 (1): 45–56. doi:10.1099/00222615-13-1-45. PMID 7359576.
- ^ a b c d e f g O'Hara, Ann M; Shanahan, Fergus (2006). "The gut flora as a forgotten organ". EMBO Reports. 7 (7): 688–93. doi:10.1038/sj.embor.7400731. PMC 1500832. PMID 16819463.
- ^ a b c d e f g h i Gibson, Glenn R. (2004). "Fibre and effects on probiotics (the prebiotic concept)". Clinical Nutrition Supplements. 1 (2): 25–31. doi:10.1016/j.clnu.2004.09.005.
- ^ a b c d e f g h i j k l Beaugerie, Laurent; Petit, Jean-Claude (2004). "Antibiotic-associated diarrhoea". Best Practice & Research Clinical Gastroenterology. 18 (2): 337–52. doi:10.1016/j.bpg.2003.10.002. PMID 15123074.
- ^ http://www.pnas.org/content/28/7/285?ijkey=931998456351a78f5d633e70cbc46b08369e0414&keytype2=tf_ipsecsha
- ^ https://www.clinicalkey.com/#!/ContentPlayerCtrl/doPlayContent/1-s2.0-S095816691200119X
- ^ a b c d e Vedantam, Gayatri; Hecht, David W (2003). "Antibiotics and anaerobes of gut origin". Current Opinion in Microbiology. 6 (5): 457–61. doi:10.1016/j.mib.2003.09.006. PMID 14572537.
- ^ Segal, Leopoldo; Blaser, Martin (2014). "A Brave New World: The Lung Microbiota in an Era of Change". Annals of the American Thoracic Society. 11: S21–S27. doi:10.1513/AnnalsATS.201306-189MG. Retrieved 2014-09-14.
- ^ Wexler, Hannah (October 2007). "Bacteroides: the Good. the Bad, and the Nitty-Gritty". Clinical Microbiology Review. 20 (4): 593–621. doi:10.1128/CMR.00008-07. PMC 2176045. PMID 17934076. Retrieved 2014-09-14.
- ^ Engel, P.; Moran, N. (2013). "The gut microbiota of insects–diversity in structure and function". FEMS Microbiology Reviews. 5: 699–735. doi:10.1111/1574-6976.12025.
- ^ Brune, A. (2014). "Symbiotic digestion of lignocellulose in termite guts". Nature Review Microbiology. 12: 168–180. doi:10.1038/nrmicro3182.
- ^ a b Dietrich, C.; Köhler, T.; Brune, A. (2014). "The cockroach origin of the termite gut microbiota: patterns in bacterial community structure reflect major evolutionary events". Applied and Environmental Microbiology. 80: 2261–2269. doi:10.1128/AEM.04206-13.
- ^ a b Mikaelyan, A.; Dietrich, C.; Köhler, T.; Poulsen, M.; Sillam-Dussès, D.; Brune, A. (2015). "Diet is the primary determinant of bacterial community structure in the guts of higher termites". Molecular Ecology. 24 (20): 5824–5895. doi:10.1111/mec.13376. PMID 26348261.
- ^ Mikaelyan, A.; Thompson, C.; Hofer, M.; Brune, A. (2016). "The deterministic assembly of complex bacterial communities in germ-free cockroach guts". Applied and Environmental Microbiology. 82: 1256–1263. doi:10.1128/AEM.03700-15.
- ^ a b Shanahan, Fergus (2002). "The host–microbe interface within the gut". Best Practice & Research Clinical Gastroenterology. 16 (6): 915–31. doi:10.1053/bega.2002.0342. PMID 12473298.
- ^ Tap, Julien; Mondot, Stanislas; Levenez, Florence; Pelletier, Eric; Caron, Christophe; Furet, Jean-Pierre; Ugarte, Edgardo; Muñoz-Tamayo, Rafael; Paslier, Denis L. E.; Nalin, Renaud; Dore, Joel; Leclerc, Marion (2009). "Towards the human intestinal microbiota phylogenetic core". Environmental Microbiology. 11 (10): 2574–84. doi:10.1111/j.1462-2920.2009.01982.x. PMID 19601958.
- ^ Khanna S, Tosh PK (January 2014). "A clinician's primer on the role of the microbiome in human health and disease". Mayo Clin. Proc. 89 (1): 107–14. doi:10.1016/j.mayocp.2013.10.011. PMID 24388028.
- ^ a b Arumugam, Manimozhiyan; Raes, Jeroen; Pelletier, Eric; Le Paslier, Denis; Yamada, Takuji; Mende, Daniel R.; Fernandes, Gabriel R.; Tap, Julien; Bruls, Thomas; Batto, Jean-Michel; Bertalan, Marcelo; Borruel, Natalia; Casellas, Francesc; Fernandez, Leyden; Gautier, Laurent; Hansen, Torben; Hattori, Masahira; Hayashi, Tetsuya; Kleerebezem, Michiel; Kurokawa, Ken; Leclerc, Marion; Levenez, Florence; Manichanh, Chaysavanh; Nielsen, H. Bjørn; Nielsen, Trine; Pons, Nicolas; Poulain, Julie; Qin, Junjie; Sicheritz-Ponten, Thomas; Tims, Sebastian (2011). "Enterotypes of the human gut microbiome". Nature. 473 (7346): 174–80. doi:10.1038/nature09944. PMC 3728647. PMID 21508958.
- ^ a b c Wu, G. D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.-Y.; Keilbaugh, S. A.; Bewtra, M.; Knights, D.; Walters, W. A.; Knight, R.; Sinha, R.; Gilroy, E.; Gupta, K.; Baldassano, R.; Nessel, L.; Li, H.; Bushman, F. D.; Lewis, J. D. (2011). "Linking Long-Term Dietary Patterns with Gut Microbial Enterotypes". Science. 334 (6052): 105–8. doi:10.1126/science.1208344. PMC 3368382. PMID 21885731.
- ^ Zimmer, Carl (April 20, 2011). "Bacteria Divide People Into 3 Types, Scientists Say". The New York Times. Retrieved April 21, 2011.
a group of scientists now report just three distinct ecosystems in the guts of people they have studied.
- ^ a b Gerritsen, Jacoline; Smidt, Hauke; Rijkers, Ger; de Vos, Willem (27 May 2011). "Intestinal microbiota in human health and disease: the impact of probiotics". Genes & Nutritions. 6 (3): 209–240. doi:10.1007/s12263-011-0229-7. PMC 3145058. PMID 21617937. Retrieved 2014-09-14.
- ^ a b c d e f g Yatsunenko, T.; Rey, F. E.; Manary, M. J.; Trehan, I.; Dominguez-Bello, M. G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R. N.; Anokhin, A. P.; Heath, A. C.; Warner, B.; Reeder, J.; Kuczynski, J.; Caporaso, J. G.; Lozupone, C. A.; Lauber, C.; Clemente, J. C.; Knights, D.; Knight, R.; Gordon, J. I. (2012). "Human gut microbiome viewed across age and geography". Nature. 486 (7402): 222–227. doi:10.1038/nature11053. PMC 3376388. PMID 22699611.
- ^ De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J. B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. (2010). "Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa". Proc. Natl. Acad. Sci. U.S.A. 107 (33): 14691–14696. doi:10.1073/pnas.1005963107. PMC 2930426. PMID 20679230.
- ^ Collado, M and Bӓuerl C et al. Defining microbiota for developing new probiotics. Microb Ecol Health Dis.2012;23 PMCID:PMC 3747743
- ^ Bettelheim, K. A.; Breadon, Alwena; Faiers, Mary C.; O'Farrell, Sheila M.; Shooter, R. A. (2009). "The origin of O serotypes of Escherichia coli in babies after normal delivery". Journal of Hygiene. 72 (1): 67–70. doi:10.1017/S0022172400023226. PMC 2130250. PMID 4593741.
- ^ a b Schwiertz, Andreas; Gruhl, Bärbel; Löbnitz, Manuela; Michel, Peter; Radke, Michael; Blaut, Michael (2003). "Development of the Intestinal Bacterial Composition in Hospitalized Preterm Infants in Comparison with Breast-Fed, Full-Term Infants". Pediatric Research. 54 (3): 393–9. doi:10.1203/01.PDR.0000078274.74607.7A. PMID 12788986.
- ^ Dominguez-Bello, Maria; Blaser, Martin; Ley, Ruth; Knight, Rob (2010). "Development of the Human Gastrointestinal Microbiota and Insights From High-Throughput Sequencing". Gastroenterology. 140 (6): 1713–1719. doi:10.1053/j.gastro.2011.02.011.
- ^ Grölund, Minna-Maija; Lehtonen, Olli-Pekka; Eerola, Erkki; Kero, Pentti (1999). "Fecal Microflora in Healthy Infants Born by Different Methods of Delivery: Permanent Changes in Intestinal Flora After Cesarean Delivery". Journal of Pediatric Gastroenterology & Nutrition. 28 (1): 19–25. doi:10.1097/00005176-199901000-00007. PMID 9890463.
- ^ MacKie, RI; Sghir, A; Gaskins, HR (1999). "Developmental microbial ecology of the neonatal gastrointestinal tract". The American Journal of Clinical Nutrition. 69 (5): 1035S–1045S. PMID 10232646.
- ^ Favier, C. F.; Vaughan, E. E.; De Vos, W. M.; Akkermans, A. D. L. (2002). "Molecular Monitoring of Succession of Bacterial Communities in Human Neonates". Applied and Environmental Microbiology. 68 (1): 219–26. doi:10.1128/AEM.68.1.219-226.2002. PMC 126580. PMID 11772630.
- ^ Coppa, Giovanni V; Bruni, Stefano; Morelli, Lorenzo; Soldi, Sara; Gabrielli, Orazio (2004). "The First Prebiotics in Humans". Journal of Clinical Gastroenterology. 38 (6 Suppl): S80–3. doi:10.1097/01.mcg.0000128926.14285.25. PMID 15220665.
- ^ Coppa, G.V.; Zampini, L.; Galeazzi, T.; Gabrielli, O. (2006). "Prebiotics in human milk: A review". Digestive and Liver Disease. 38: S291–4. doi:10.1016/S1590-8658(07)60013-9. PMID 17259094.
- ^ Harmsen, Hermie J. M.; Wildeboer-Veloo, Alida C. M.; Raangs, Gerwin C.; Wagendorp, Arjen A.; Klijn, Nicolette; Bindels, Jacques G.; Welling, Gjalt W. (2000). "Analysis of Intestinal Flora Development in Breast-Fed and Formula-Fed Infants by Using Molecular Identification and Detection Methods". Journal of Pediatric Gastroenterology and Nutrition. 30 (1): 61–7. doi:10.1097/00005176-200001000-00019. PMID 10630441.
- ^ Fanaro, S; Chierici, R; Guerrini, P; Vigi, V (2003). "Intestinal microflora in early infancy: Composition and development". Acta paediatrica. 91 (441): 48–55. PMID 14599042.
- ^ a b c Wynne, Anthony G; McCartney, Anne L; Brostoff, Jonathan; Hudspith, Barry N; Gibson, Glenn R (2004). "An in vitro assessment of the effects of broad-spectrum antibiotics on the human gut microflora and concomitant isolation of a Lactobacillus plantarum with anti-Candida activities". Anaerobe. 10 (3): 165–9. doi:10.1016/j.anaerobe.2004.03.002. PMID 16701514.
- ^ a b c Keeley J. 2004. Good bacteria trigger proteins to protect the gut. Howard Hughes Medical Institute. EurekAlert. Accessed January 9, 2007.
- ^ Jewell, A.P. (2005). "Is the liver an important site for the development of immune tolerance to tumours?". Medical Hypotheses. 64 (4): 751–4. doi:10.1016/j.mehy.2004.10.002. PMID 15694692.
- ^ Tlaskalová-Hogenová H., Stepánková R., Hudcovic T., Tucková L., Cukrowska B., Lodinová-Zádníková R., Kozáková H., Rossmann P., Bártová J., Sokol D.; et al. (2004). "Commensal bacteria (normal microflora), mucosal immunity and chronic inflammatory and autoimmune diseases". Immunol. Lett. 93: 97–108. doi:10.1016/j.imlet.2004.02.005.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Penders J., Stobberingh E. E., van den Brandt P. A., Thijs C. (2007). "The role of the intestinal microbiota in the development of atopic disorders". Allergy. 62: 1223–36. doi:10.1111/j.1398-9995.2007.01462.x.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Björkstén B., Sepp E., Julge K., Voor T., Mikelsaar M. (2001). "Allergy development and the intestinal microflora during the first year of life". J. Allergy Clin. Immunol. 108: 516–20. doi:10.1067/mai.2001.118130. PMID 11590374.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Trompette A, Gollwitzer ES, Yadava K, Sichelstiel AK, Sprenger N, Ngom-Bru C, Blanchard C, Junt T, Nicod LP, Harris NL, Marsland BJ (2014). "Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis". Nat. Med. 20 (2): 159–66. doi:10.1038/nm.3444. PMID 24390308.
- ^ Mantis, N J; Rol, N; Corthésy, B (2011). "Secretory IgA's complex roles in immunity and mucosal homeostasis in the gut". Mucosal Immunology. 4 (6): 603–11. doi:10.1038/mi.2011.41. PMC 3774538. PMID 21975936.
- ^ a b c d e f g h Guarner, Francisco; Malagelada, Juan-R (2003). "Role of bacteria in experimental colitis". Best Practice & Research Clinical Gastroenterology. 17 (5): 793–804. doi:10.1016/S1521-6918(03)00068-4. PMID 14507589.
- ^ Kim, Eun-Hee; Hong, Hua; Choi, Ki-Seok; Han, Young-Min; Kangwan, Napapan; Cho, Young Chae; Hahm, Ki Baik (April 2012). "High Concentrated Probiotics Improve Inflammatory Bowel Diseases Better than Commercial Concentration of Probiotics". Journal of Food & Drug Analysis. 20: 292–5.
- ^ a b c Carman, Robert J.; Simon, Mary Alice; Fernández, Haydée; Miller, Margaret A.; Bartholomew, Mary J. (2004). "Ciprofloxacin at low levels disrupts colonization resistance of human fecal microflora growing in chemostats". Regulatory Toxicology and Pharmacology. 40 (3): 319–26. doi:10.1016/j.yrtph.2004.08.005. PMID 15546686.
- ^ Brandt, Lawrence J.; Borody, Thomas Julius; Campbell, Jordana (2011). "Endoscopic Fecal Microbiota Transplantation". Journal of Clinical Gastroenterology. 45 (8): 655–7. doi:10.1097/MCG.0b013e3182257d4f. PMID 21716124.
- ^ Knight, DJW; Girling, KJ (2003). "Gut flora in health and disease". The Lancet. 361 (9371): 512–9. doi:10.1016/S0140-6736(03)13438-1. PMID 12781578.
- ^ a b c d Cho, I.; Yamanishi, S.; Cox, L.; Methé, B. A.; Zavadil, J.; Li, K.; Gao, Z.; Mahana, D.; Raju, K.; Teitler, I.; Li, H.; Alekseyenko, A. V.; Blaser, M. J. (2012). "Antibiotics in early life alter the murine colonic microbiome and adiposity". Nature. 488 (7413): 621–6. doi:10.1038/nature11400. PMC 3553221. PMID 22914093.
- ^ Baker, Monya (2012). "Pregnancy alters resident gut microbes". Nature. doi:10.1038/nature.2012.11118.
- ^ a b Ley, Ruth E.; Turnbaugh, Peter J.; Klein, Samuel; Gordon, Jeffrey I. (2006). "Microbial ecology: Human gut microbes associated with obesity". Nature. 444 (7122): 1022–3. doi:10.1038/4441022a. PMID 17183309.
- ^ Hold, Georgina L.; Pryde, Susan E.; Russell, Valerie J.; Furrie, Elizabeth; Flint, Harry J. (2002). "Assessment of microbial diversity in human colonic samples by 16S rDNA sequence analysis". FEMS Microbiology Ecology. 39 (1): 33–9. doi:10.1111/j.1574-6941.2002.tb00904.x. PMID 19709182.
- ^ "Health effects of artificial sweeteners". CNN. Retrieved 2016-03-24.
- ^ "Bacteria in your gut: 4 healthy habits - CNN.com". CNN. Retrieved 2016-03-24.
- ^ Abou-Donia, Mohamed B.; El-Masry, Eman M.; Abdel-Rahman, Ali A.; McLendon, Roger E.; Schiffman, Susan S. (2008-01-01). "Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome p-450 in male rats". Journal of Toxicology and Environmental Health. Part A. 71 (21): 1415–1429. doi:10.1080/15287390802328630. ISSN 1528-7394. PMID 18800291.
- ^ Omran, Arthur (10 November 2013). "Metabolic Effects of Sucralose on Environmental Bacteria". Hindawi Publishing Corporation. Journal of Toxicology. Retrieved 21 March 2016.
- ^ a b c d Suenaert, Peter; Bulteel, Veerle; Lemmens, Liesbeth; Noman, Maja; Geypens, Benny; Van Assche, Gert Van; Geboes, Karel; Ceuppens, Jan L.; Rutgeerts, Paul (2002). "Anti-tumor necrosis factor treatment restores the gut barrier in Crohn's disease". The American Journal of Gastroenterology. 97 (8): 2000–4. doi:10.1111/j.1572-0241.2002.05914.x. PMID 12190167.
- ^ Garcia-Tsao, Guadalupe; Wiest, Reiner (2004). "Gut microflora in the pathogenesis of the complications of cirrhosis". Best Practice & Research Clinical Gastroenterology. 18 (2): 353–72. doi:10.1016/j.bpg.2003.10.005. PMID 15123075.
- ^ a b c Hugot, Jean-Pierre (2004). "Inflammatory bowel disease: A complex group of genetic disorders". Best Practice & Research Clinical Gastroenterology. 18 (3): 451–62. doi:10.1016/j.bpg.2004.01.001. PMID 15157820.
- ^ a b Veltkamp, Claudia; Tonkonogy, Susan L.; De Jong, Ype P.; Albright, Carol; Grenther, Wetonia B.; Balish, Edward; Terhorst, Cox; Sartor, R.Balfour (2001). "Continuous stimulation by normal luminal bacteria is essential for the development and perpetuation of colitis in Tgϵ26 mice". Gastroenterology. 120 (4): 900–13. doi:10.1053/gast.2001.22547. PMID 11231944.
- ^ Wendelsdorf, Katherine. (2013). "Gut microbiota from twins discordant for obesity modulate metabolism in mice". Science. 341 (6150): 1241214. doi:10.1126/science.1241214.
- ^ Turnbaugh, Peter J.; Ley, Ruth E.; Mahowald, Michael A.; Magrini, Vincent; Mardis, Elaine R.; Gordon, Jeffrey I. (2006). "An obesity-associated gut microbiome with increased capacity for energy harvest". Nature. 444 (7122): 1027–31. doi:10.1038/nature05414. PMID 17183312.
- ^ Backhed, F.; Manchester, J. K.; Semenkovich, C. F.; Gordon, J. I. (2007). "Mechanisms underlying the resistance to diet-induced obesity in germ-free mice". Proceedings of the National Academy of Sciences. 104 (3): 979–984. doi:10.1073/pnas.0605374104. PMC 1764762. PMID 17210919.
- ^ Backhed, F.; Manchester, JK; Semenkovich, CF; Gordon, JI (2004). "The gut microbiota as an environmental factor that regulates fat storage". Proceedings of the National Academy of Sciences. 101 (44): 979–84. doi:10.1073/pnas.0407076101. PMC 1764762. PMID 17210919.
- ^ Walker, A. W.; Parkhill, J. (2013). "Fighting Obesity with Bacteria". Scrience. 341 (6150): 1069–1070. doi:10.1126/science.1243787.
Further reading
- Books
- Hattner, Jo Ann Tatum; Anderes, Susan (2009). Gut Insight: probiotics and prebiotics for digestive health and well-being. Hattner Nutrition. ISBN 978-0-578-02615-2.
- Review articles
- Maranduba, CM; De Castro, SB; de Souza, GT; Rossato, C; da Guia, FC; Valente, MA; Rettore, JV; Maranduba, CP; de Souza, CM; do Carmo, AM; Macedo, GC; Silva, FS (2015). "Intestinal Microbiota as Modulators of the Immune System and Neuroimmune System: Impact on the Host Health and Homeostasis". Journal of Immunology Research. 2015: 931574. doi:10.1155/2015/931574. PMC 4352473. PMID 25759850.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - Prakash, Satya; Tomaro-Duchesneau, Catherine; Saha, Shyamali; Cantor, Arielle (2011). "The Gut Microbiota and Human Health with an Emphasis on the Use of Microencapsulated Bacterial Cells". Journal of Biomedicine and Biotechnology. 2011: 1–12. doi:10.1155/2011/981214. PMC 3134400. PMID 21772792.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - De Preter, Vicky; Hamer, Henrike M; Windey, Karen; Verbeke, Kristin (2011). "The impact of pre- and/or probiotics on human colonic metabolism: Does it affect human health?". Molecular Nutrition & Food Research. 55 (1): 46–57. doi:10.1002/mnfr.201000451. PMID 21207512.
- Prakash, Satya; Rodes, Laetitia; Coussa-Charley, Michael; Tomaro-Duchesneau, Catherine; Tomaro-Duchesneau, Catherine; Coussa-Charley; Rodes (2011). "Gut microbiota: Next frontier in understanding human health and development of biotherapeutics". Biologics: Targets and Therapy. 5: 71–86. doi:10.2147/BTT.S19099. PMC 3156250. PMID 21847343.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - Wu, G. D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.-Y.; Keilbaugh, S. A.; Bewtra, M.; Knights, D.; Walters, W. A.; Knight, R.; Sinha, R.; Gilroy, E.; Gupta, K.; Baldassano, R.; Nessel, L.; Li, H.; Bushman, F. D.; Lewis, J. D. (2011). "Linking Long-Term Dietary Patterns with Gut Microbial Enterotypes". Science. 334 (6052): 105–8. doi:10.1126/science.1208344. PMC 3368382. PMID 21885731.
