Allotropes of plutonium
| Phase | Crystal Structure | Density (g/cm3) |
|---|---|---|
| alpha ( | simple monoclinic | 19.86 |
| beta ( | body-centered monoclinic | 17.70 |
| gamma ( | face-centered orthorhombic | 17.14 |
| delta ( | face-centered cubic | 15.92 |
| delta prime ( | body-centered tetragonal | 16.00 |
| epsilon ( | body-centered cubic | 16.51 |
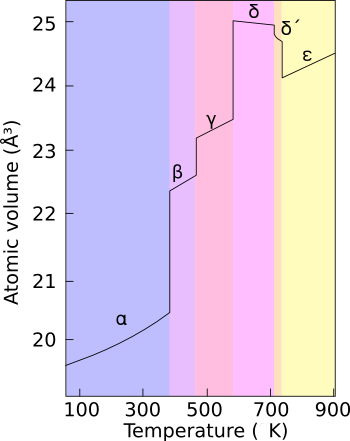
Plutonium occurs in a variety of allotropes, even at ambient pressure. These allotropes differ widely in crystal structure and density; the
Overview
[edit]Plutonium normally has six allotropes and forms a seventh (zeta,
Machining plutonium
[edit]The presence of these many allotropes makes machining plutonium very difficult, as it changes state very readily. For example, the alpha (
The reasons for the complicated phase diagram are not entirely understood; recent research has focused on constructing accurate computer models of the phase transitions. The
Stabilization
[edit]Plutonium in the delta (
Gallium, aluminium, americium, scandium and cerium can stabilize the


References
[edit]- ^ a b c Baker, Richard D.; Hecker, Siegfried S.; Harbur, Delbert R. (Winter–Spring 1983). "Plutonium: A Wartime Nightmare but a Metallurgist's Dream" (PDF). Los Alamos Science. Los Alamos National Laboratory: 148, 150–151.
- ^ a b Hecker, Siegfried S. (2000). "Plutonium and its alloys: from atoms to microstructure" (PDF). Los Alamos Science. 26: 290–335.
- ^ Miner, William N.; Schonfeld, Fred W. (1968). "Plutonium". In Clifford A. Hampel (ed.). The Encyclopedia of the Chemical Elements. New York: Reinhold Book Corporation. p. 544.
- ^ Plutonium Crystal Phase Transitions. Globalsecurity.org (27 April 2005). Retrieved 2010-02-08.
- ^ David A. Young (11 September 1975). "Phase Diagrams of the Elements" (PDF). Lawrence Livermore Laboratory.
