Internexin
| Internexin neuronal intermediate filament protein, alpha | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | INA | ||||||
| Alt. symbols | NEF5 | ||||||
| NCBI gene | 9118 | ||||||
| HGNC | 6057 | ||||||
| OMIM | 605338 | ||||||
| RefSeq | NM_032727 | ||||||
| UniProt | Q16352 | ||||||
| Other data | |||||||
| Locus | Chr. 10 q24 | ||||||
| |||||||
Internexin, alpha-internexin, is a Class IV intermediate filament approximately 66 KDa. The protein was originally purified from rat optic nerve and spinal cord.[1] The protein copurifies with other neurofilament subunits, as it was originally discovered, however in some mature neurons it can be the only neurofilament expressed. The protein is present in developing neuroblasts and in the central nervous system of adults. The protein is a major component of the intermediate filament network in small interneurons and cerebellar granule cells, where it is present in the parallel fibers.
Structure
[edit]Alpha-internexin has a homologous central rod domain of approximately 310 amino acid residues that form a highly conserved alpha helical region. The central rod domain is responsible for coiled-coil structure and is flanked by an amino terminal head region and a carboxy terminal tail.[2] This rod domain is also involved in the 10 nm filament assembly structure. The head and tail regions contain segments that are highly homologous to the NF-M’s structure.[1] The head region is highly basic and contains many serine and threonine polymers while the tail region has distinct sequence motifs like a glutamate rich region.[3] The alpha domain is composed of heptad repeats of hydrophobic residues that aid the formation of a coiled coil structure.[3] The structure of Alpha-internexin is highly conserved between rats, mice and humans.[1]
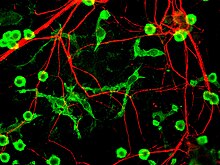
Alpha-internexin can form homopolymers, unlike the heteropolymer the neurofilaments form. This formation suggests that
- Step 1: in the first step of IF assembly two parallel, unstaggered intermediate filament polypeptides chains form a dimer via their a-helical rod domains; these dimers can be either homodimers or heterodimers.
- Step 2: the dimers may associate laterally to form antiparallel, unstaggered tetramers or antiparallel, staggered tetramers.
- Step 3: the dimers may also associate longitudinally with a short head-to-tail overlap of the a-helical rod domains.
- Step 4: these lateral and longitudinal associations lead to the formation of protofibrils (octamers) and ultimately 10 nm intermediate filaments.[5]
The close connection between the neurofilament triplet proteins and
Function and expression
[edit]It is expressed in early development in the neuroblast along with
In adult cells,
Alpha-internexin is a brain and central nervous system filament that is involved in neuronal development and has been suggested to play a role in axonal outgrowth. Gefiltin and xefiltin, homologs of
Disease associations
[edit]See also
[edit]References
[edit]- ^ a b c d e f Levavasseur F, Zhu Q, and JP Julien. No requirement of alpha-internexin for nervous system development and for radial growth of axons. Molecular Brain Research. 69:104-112. (1999).
- ^ Lariviere, R. and JP Julien. Functions of Intermediate Filaments in Neuronal Development and Disease. Journal of Neurobiology. 58(1): 131-48. (2004).
- ^ a b c Catalogue# CPCA-a-Int: Chicken Polyclonal Antibody to alpha-internexin. EnCor Biotechnology Inc. 2011.
- ^ a b c d e f Duprey, P and D. Paulin. What can be learned from intermediate filament gene regulation in the mouse embryo? International Journal of Developmental Biology. 39:443-457. (1995).
- ^ Ching G and R. Liem. Analysis of roles of the head domains of type IV rat neuronal intermediate filament proteins in filament assembly using domain-swapped chimeric proteins. Journal of Cell Science. 112:2233-2240. (1999).
External links
[edit]- Interactions of internexin alpha
- alpha-internexin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
