Betaine

A betaine (/ˈbiːtə.iːn, bɪˈteɪ-, -ɪn/) in chemistry is any neutral chemical compound with a positively charged cationic functional group that bears no hydrogen atom, such as a quaternary ammonium or phosphonium cation (generally: onium ions), and with a negatively charged functional group, such as a carboxylate group that may not be adjacent to the cationic site.[1] Historically, the term was reserved for trimethylglycine (TMG), which is involved in methylation reactions and detoxification of homocysteine.[1] This is a modified amino acid consisting of glycine with three methyl groups serving as methyl donor for various metabolic pathways.[2]
Pronunciation
[edit]The pronunciation of the compound reflects its origin and first isolation from sugar beets (Beta vulgaris subsp. vulgaris), and does not derive from the Greek letter beta (
Glycine betaine
[edit]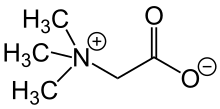
The original betaine, N,N,N-trimethylglycine, was named after its discovery in sugar beet (Beta vulgaris subsp. vulgaris) in the nineteenth century.[4] It is a small N-trimethylated amino acid. It is a zwitterion, which cannot isomerize because there is no labile hydrogen atom attached to the nitrogen atom. This substance may be called glycine betaine to distinguish it from other betaines.
Uses
[edit]Biochemistry
[edit]Phosphonium betaines are intermediates in the Wittig reaction. The addition of betaine to polymerase chain reactions improves the amplification of DNA by reducing the formation of secondary structure in GC-rich regions. The addition of betaine may enhance the specificity of the polymerase chain reaction by eliminating the base pair composition dependence of DNA melting.[5][6]
Food additive
[edit]In 2017, the European Food Safety Authority concluded that betaine was safe "as a novel food to be used at a maximum intake level of 6 mg/kg body weight per day in addition to the intake from the background diet."[7]
Approved drug
[edit]A prescription drug (Cystadane)[8] containing betaine has limited use for oral treatment of genetic homocystinuria to lower levels of homocysteine in circulating blood.[2][1]
Dietary supplement
[edit]Trimethylglycine, a betaine, is used as a dietary supplement, although there is no evidence that it is effective or safe.[9] Common side effects of taking oral betaine include nausea and stomach upset.[8]
Safety
[edit]Many betaines are irritants of the eyes and skin.[1]
See also
[edit]References
[edit]- ^ a b c d "Betaine". PubChem, US National Library of Medicine. 21 August 2021. Retrieved 26 August 2021.
- ^ a b "Betaine". LiverTox, US National Library of Medicine. 26 September 2017. PMID 31644082. Retrieved 26 August 2021.
- ^ Alex Nickon and Ernest F. Silversmith (1987). Organic Chemistry, the Name Game: Modern Coined Terms and Their Origins. Pergamon. ISBN 978-0080344812.
- ^ DNA Methylation and Complex Human Disease, Michel Neidhart
- ^ Rees, William A.; Yager, Thomas D.; Korte, John; Von Hippel, Peter H. (1993). "Betaine can eliminate the base pair composition dependence of DNA melting". Biochemistry. 32 (1): 137–44. doi:10.1021/bi00052a019. PMID 8418834.
- ^ Henke, W; Herdel, K; Jung, K; Schnorr, D; Loening, SA (1997). "Betaine improves the PCR amplification of GC-rich DNA sequences". Nucleic Acids Research. 25 (19): 3957–8. doi:10.1093/nar/25.19.3957. PMC 146979. PMID 9380524.
- ^ EFSA Panel on Nutrition, Novel Foods and Food Allergens (5 April 2019). "Safety of betaine as a novel food pursuant to Regulation (EU) 2015/2283". EFSA Journal. 17 (4): e05658. doi:10.2903/j.efsa.2019.5658. PMC 7009179. PMID 32626284.
- ^ a b "Betaine". Drugs.com. 11 November 2020. Retrieved 26 August 2021.
- ^ Van Every, Derrick W.; Plotkin, Daniel L.; Delcastillo, Kenneth; Cholewa, Jason; Schoenfeld, Brad J. (2021-08-01). "Betaine Supplementation: A Critical Review of Its Efficacy for Improving Muscle Strength, Power, and Body Composition". Strength & Conditioning Journal. 43 (4): 53–61. doi:10.1519/SSC.0000000000000622. ISSN 1524-1602. S2CID 233966447.
Further reading
[edit]- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "betaines". doi:10.1351/goldbook.B00637
