WB-4101
Appearance
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
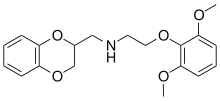
| Formula | C19H23NO5 |
| Molar mass | 345.395 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
WB-4101 is a compound which acts as an antagonist at the
See also
[edit]References
[edit]- ^ Green PN, Shapero M, Wilson C (March 1969). "The synthesis and pharmacological properties of a series of 2-substituted aminomethyl-1,4-benzodioxanes". Journal of Medicinal Chemistry. 12 (2): 326–9. doi:10.1021/jm00302a033. PMID 5791620.
- ^ Morrow AL, Creese I (April 1986). "Characterization of alpha 1-adrenergic receptor subtypes in rat brain: a reevaluation of [3H]WB4104 and [3H]prazosin binding". Molecular Pharmacology. 29 (4): 321–30. PMID 3010073.
- ^ Melchiorre C, Bolognesi ML, Budriesi R, Chiarini A, Giardinà D, Minarini A, et al. (April 1998). "Search for selective antagonists at alpha 1-adrenoreceptors: neutral or negative antagonism?". Farmaco. 53 (4): 278–86. doi:10.1016/S0014-827X(98)00022-6. PMID 9658586.
- ^ Melchiorre C, Angeli P, Bolognesi ML, Chiarini A, Giardinà D, Gulini U, et al. (March 2000). "Alpha 1-adrenoreceptor antagonists bearing a quinazoline or a benzodioxane moiety". Pharmaceutica Acta Helvetiae. 74 (2–3): 181–90. doi:10.1016/S0031-6865(99)00049-7. PMID 10812956.
- ^ Fumagalli L, Pallavicini M, Budriesi R, Gobbi M, Straniero V, Zagami M, et al. (December 2012). "Affinity and activity profiling of unichiral 8-substituted 1,4-benzodioxane analogues of WB4101 reveals a potent and selective
α 1B-adrenoceptor antagonist". European Journal of Medicinal Chemistry. 58: 184–91. doi:10.1016/j.ejmech.2012.09.049. PMID 23124215. - ^ Fumagalli L, Pallavicini M, Budriesi R, Bolchi C, Canovi M, Chiarini A, et al. (August 2013). "6-methoxy-7-benzofuranoxy and 6-methoxy-7-indolyloxy analogues of 2-[2-(2,6-Dimethoxyphenoxy)ethyl]aminomethyl-1,4-benzodioxane (WB4101):1 discovery of a potent and selective
α 1D-adrenoceptor antagonist". Journal of Medicinal Chemistry. 56 (16): 6402–12. doi:10.1021/jm400867d. PMID 23902232.
