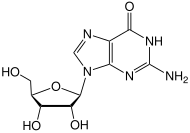
Guanosine (symbol G or Guo) is a purine nucleoside comprising guanine attached to a ribose (ribofuranose) ring via a
| |

| |
| Names | |
|---|---|
| IUPAC name
Guanosine[1]
| |
| Systematic IUPAC name
2-Amino-9-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,9-dihydro-6H-purin-6-one | |
| Other names
Guanine riboside
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.844 |
| KEGG | |
| MeSH | Guanosine |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H13N5O5 | |
| Molar mass | 283.241 |
| Appearance | white, crystalline powder[2] |
| Odor | odorless[2] |
| Melting point | 239 (decomposes)[3] |
| -149.1·10−6 cm3/mol | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Physical and chemical properties
editGuanosine is a white, crystalline powder with no odor and mild saline taste.[2] It is very soluble in acetic acid, slightly soluble in water, insoluble in ethanol, diethyl ether, benzene and chloroform.[3]
Functions
editGuanosine is required for an RNA splicing reaction in mRNA, when a "self-splicing" intron removes itself from the mRNA message by cutting at both ends, re-ligating, and leaving just the exons on either side to be translated into protein.[4]
Uses
editThe antiviral drug acyclovir, often used in herpes treatment, and the anti-HIV drug abacavir, are structurally similar to guanosine.[5][6] Guanosine was also used to make regadenoson.
Sources
editGuanosine can be found in pancreas, clover, coffee plant, and pollen of pines.[2]
References
edit- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 1421. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ a b c d Robert A. Lewis, Michael D. Larrañaga, Richard J. Lewis Sr. (2016). Hawley's Condensed Chemical Dictionary (16th ed.). Hoboken, New Jersey: John Wiley & Sons, Inc. p. 688. ISBN 978-1-118-13515-0.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ a b William M. Haynes (2016). CRC Handbook of Chemistry and Physics (97th ed.). Boca Raton: CRC Press. pp. 3–286. ISBN 978-1-4987-5429-3.
- ^ Splicing (JPG) Archived June 13, 2010, at the Wayback Machine
- ^ "Acyclovir". The American Society of Health-System Pharmacists. Archived from the original on 2015-01-05. Retrieved Jan 1, 2015.
- ^ Product Information: ZIAGEN(R) oral tablets, oral solution, abacavir sulfate oral tablets, oral solution. ViiV Healthcare (per Manufacturer), Research Triangle Park, NC, 2015.