From Wikipedia, the free encyclopedia
3α あるふぁ
Names
IUPAC name
5α あるふぁ α あるふぁ β べーた
Systematic IUPAC name
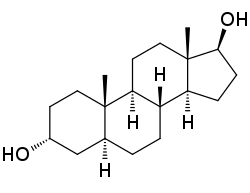
(1S ,3aS ,3bR ,5aS ,7R ,9aS ,9bS ,11aS )-9a,11a-Dimethylhexadecahydro-1H -cyclopenta[a ]phenanthrene-1,7-diol
Other names
Hombreol
Identifiers
ChEBI
ChemSpider
ECHA InfoCard 100.015.862
UNII
InChI=1S/C19H32O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h12-17,20-21H,3-11H2,1-2H3/t12-,13+,14-,15-,16-,17-,18-,19-/m0/s1
Key: CBMYJHIOYJEBSB-KHOSGYARSA-N
InChI=1/C19H32O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h12-17,20-21H,3-11H2,1-2H3/t12-,13+,14-,15-,16-,17-,18-,19-/m0/s1
Key: CBMYJHIOYJEBSB-KHOSGYARBM
C[C@]12CC[C@H]3[C@@H](CC[C@H]4C[C@H](O)CC[C@@]43C)[C@@H]1CC[C@@H]2O
Properties
C 19 H 32 O 2
Molar mass
−1
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
3α あるふぁ also known as 5α あるふぁ α あるふぁ β べーた and sometimes shortened in the literature to 3α あるふぁ , is an endogenous steroid hormone and neurosteroid and a metabolite of androgens like dihydrotestosterone (DHT).[ 1] [ 2] [ 3]
Biological activity [ edit ] 3α あるふぁ inhibitory androstane neurosteroid and weak androgen and estrogen .[ 1] [ 2] [ 3]
As a neurosteroid, it acts as a potent positive allosteric modulator of the GABAA receptor ,[ 4] rewarding ,[ 5] [ 6] anxiolytic ,[ 7] pro-sexual ,[ 8] anticonvulsant effects.[ 9] [ 10] testosterone and DHT are known to have many of the same effects as 3α あるふぁ in vivo , it is thought that this compound may in part be responsible for said effects.[ 5] [ 6] [ 7] [ 10]
Relative to its isomer 3β べーた , which is a potent estrogen , 3α あるふぁ affinity for the estrogen receptors , with a several-fold preference for ERβ べーた over ERα あるふぁ .[ 11] [ 12] affinity of estradiol at the ERα あるふぁ β べーた [ 13]
3α あるふぁ sex hormone-binding globulin (SHBG), similar to that of testosterone .[ 14]
3α あるふぁ α あるふぁ α あるふぁ β べーた naturally occurring androstane steroid and a structural analogue of DHT (5α あるふぁ β べーた positional isomer of 3α あるふぁ 3β べーた .
An orally active synthetic analogue of 3α あるふぁ 17α あるふぁ α あるふぁ (HE-3235, Apoptone), was formerly under investigation for the treatment of prostate cancer and breast cancer .[ 15]
^ a b Reddy DS (2010). "Neurosteroids". Sex Differences in the Human Brain, their Underpinnings and Implications . Progress in Brain Research. Vol. 186. pp. 113–37. doi :10.1016/B978-0-444-53630-3.00008-7 . ISBN 9780444536303 PMC 3139029 PMID 21094889 . ^ a b Jin Y, Penning TM (March 2001). "Steroid 5alpha-reductases and 3alpha-hydroxysteroid dehydrogenases: key enzymes in androgen metabolism". Best Pract. Res. Clin. Endocrinol. Metab . 15 (1): 79–94. doi :10.1053/beem.2001.0120 . PMID 11469812 . ^ a b Penning TM, Bauman DR, Jin Y, Rizner TL (February 2007). "Identification of the molecular switch that regulates access of 5alpha-DHT to the androgen receptor" . Mol. Cell. Endocrinol . 265–266: 77–82. doi :10.1016/j.mce.2006.12.007 . PMC 1857325 PMID 17223255 . ^ Reddy DS, Jian K (September 2010). "The testosterone-derived neurosteroid androstanediol is a positive allosteric modulator of GABAA receptors" . J. Pharmacol. Exp. Ther . 334 (3): 1031–41. doi :10.1124/jpet.110.169854 . PMC 2939675 PMID 20551294 . ^ a b Frye CA (February 2007). "Some rewarding effects of androgens may be mediated by actions of its 5alpha-reduced metabolite 3alpha-androstanediol" . Pharmacol. Biochem. Behav . 86 (2): 354–67. doi :10.1016/j.pbb.2006.10.003 . PMC 1857333 PMID 17112575 . ^ a b Rosellini RA, Svare BB, Rhodes ME, Frye CA (November 2001). "The testosterone metabolite and neurosteroid 3alpha-androstanediol may mediate the effects of testosterone on conditioned place preference". Brain Res. Brain Res. Rev . 37 (1–3): 162–71. doi :10.1016/s0165-0173(01)00116-3 . PMID 11744084 . S2CID 44735355 . ^ a b Fernández-Guasti A, Martínez-Mota L (September 2005). "Anxiolytic-like actions of testosterone in the burying behavior test: role of androgen and GABA-benzodiazepine receptors". Psychoneuroendocrinology . 30 (8): 762–70. doi :10.1016/j.psyneuen.2005.03.006 . PMID 15919582 . S2CID 3150411 . ^ Sánchez Montoya EL, Hernández L, Barreto-Estrada JL, Ortiz JG, Jorge JC (November 2010). "The testosterone metabolite 3α あるふぁ . J Sex Med . 7 (11): 3598–609. doi :10.1111/j.1743-6109.2010.01937.x . PMC 4360968 PMID 20646182 . ^ Reddy DS (March 2004). "Anticonvulsant activity of the testosterone-derived neurosteroid 3alpha-androstanediol". NeuroReport . 15 (3): 515–8. doi :10.1097/00001756-200403010-00026 . PMID 15094514 . S2CID 29967602 . ^ a b Reddy DS (2004). "Testosterone modulation of seizure susceptibility is mediated by neurosteroids 3alpha-androstanediol and 17beta-estradiol". Neuroscience . 129 (1): 195–207. doi :10.1016/j.neuroscience.2004.08.002 . PMID 15489042 . S2CID 54391883 . ^ Baker ME (2002). "Recent insights into the origins of adrenal and sex steroid receptors" (PDF) . J. Mol. Endocrinol . 28 (3): 149–52. doi :10.1677/jme.0.0280149 PMID 12063181 . ^ Kuiper, George G. J. M.; Carlsson, Bo; Grandien, Kaj; Enmark, Eva; Häggblad, Johan; Nilsson, Stefan; Gustafsson, Jan-Åke (1997). "Comparison of the Ligand Binding Specificity and Transcript Tissue Distribution of Estrogen Receptors α あるふぁ β べーた . Endocrinology . 138 (3): 863–870. doi :10.1210/endo.138.3.4979 ISSN 0013-7227 . PMID 9048584 . ^ Kuiper GG, Carlsson B, Grandien K, Enmark E, Häggblad J, Nilsson S, Gustafsson JA (1997). "Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta" . Endocrinology . 138 (3): 863–70. doi :10.1210/endo.138.3.4979 PMID 9048584 . ^ Hong H, Branham WS, Ng HW, Moland CL, Dial SL, Fang H, Perkins R, Sheehan D, Tong W (February 2015). "Human sex hormone-binding globulin binding affinities of 125 structurally diverse chemicals and comparison with their binding to androgen receptor, estrogen receptor, and α あるふぁ . Toxicol. Sci . 143 (2): 333–48. doi :10.1093/toxsci/kfu231 PMID 25349334 . ^ Ahlem C, Kennedy M, Page T, Bell D, Delorme E, Villegas S, Reading C, White S, Stickney D, Frincke J (2012). "17α あるふぁ α あるふぁ β べーた Invest New Drugs . 30 (1): 59–78. doi :10.1007/s10637-010-9517-0 . PMID 20814732 . S2CID 24785562 .
AR Tooltip Androgen receptor
Agonists SARMs Tooltip Selective androgen receptor modulator Antagonists
GPRC6A
ER Tooltip Estrogen receptor
Agonists
Steroidal: 2-Hydroxyestradiol 2-Hydroxyestrone 3-Methyl-19-methyleneandrosta-3,5-dien-17β べーた 3α あるふぁ 3α あるふぁ α あるふぁ 3β べーた α あるふぁ 3α あるふぁ 3β べーた 3β べーた 4-Androstenediol 4-Androstenedione 4-Fluoroestradiol 4-Hydroxyestradiol 4-Hydroxyestrone 4-Methoxyestradiol 4-Methoxyestrone 5-Androstenediol 7-Oxo-DHEA 7α あるふぁ 7α あるふぁ 7β べーた 8,9-Dehydroestradiol 8,9-Dehydroestrone 8β べーた 10β べーた β べーた 11β べーた 11β べーた 15α あるふぁ 16-Ketoestradiol 16-Ketoestrone 16α あるふぁ 16α あるふぁ 16α あるふぁ 16α あるふぁ 16α あるふぁ 16β べーた 16β べーた α あるふぁ β べーた α あるふぁ 17α あるふぁ (alfatradiol )17α あるふぁ 17α あるふぁ 17α あるふぁ α あるふぁ α あるふぁ 17α あるふぁ α あるふぁ 17α あるふぁ β べーた 17β べーた 17β べーた 17β べーた α あるふぁ Abiraterone Abiraterone acetate Alestramustine Almestrone Anabolic steroids (e.g., testosterone and esters , methyltestosterone , metandienone (methandrostenolone) , nandrolone and esters , many others; via estrogenic metabolites)Atrimustine Bolandiol Bolandiol dipropionate Butolame Clomestrone Cloxestradiol
Conjugated estriol Conjugated estrogens Cyclodiol Cyclotriol DHEA DHEA-S ent -EstradiolEpiestriol (16β べーた β べーた β べーた Epimestrol Equilenin Equilin ERA-63 (ORG-37663) Esterified estrogens Estetrol Estradiol
Estramustine Estramustine phosphate Estrapronicate Estrazinol Estriol
Estrofurate Estrogenic substances Estromustine Estrone
Etamestrol (eptamestrol) Ethinylandrostenediol
Ethinylestradiol
Ethinylestriol Ethylestradiol Etynodiol Etynodiol diacetate Hexolame Hippulin Hydroxyestrone diacetate Lynestrenol Lynestrenol phenylpropionate Mestranol Methylestradiol Moxestrol Mytatrienediol Nilestriol Norethisterone Noretynodrel Orestrate Pentolame Prodiame Prolame Promestriene RU-16117 Quinestradol Quinestrol Tibolone Xenoestrogens: Anise -related (e.g., anethole , anol , dianethole , dianol , photoanethole )Chalconoids (e.g., isoliquiritigenin , phloretin , phlorizin (phloridzin) , wedelolactone )Coumestans (e.g., coumestrol , psoralidin )Flavonoids (incl. 7,8-DHF , 8-prenylnaringenin , apigenin , baicalein , baicalin , biochanin A , calycosin , catechin , daidzein , daidzin , ECG , EGCG , epicatechin , equol , formononetin , glabrene , glabridin , genistein , genistin , glycitein , kaempferol , liquiritigenin , mirificin , myricetin , naringenin , penduletin , pinocembrin , prunetin , puerarin , quercetin , tectoridin , tectorigenin )Lavender oil Lignans (e.g., enterodiol , enterolactone , nyasol (cis -hinokiresinol) )Metalloestrogens (e.g., cadmium )Pesticides (e.g., alternariol , dieldrin , endosulfan , fenarimol , HPTE , methiocarb , methoxychlor , triclocarban , triclosan )Phytosteroids (e.g., digitoxin (digitalis ), diosgenin , guggulsterone )Phytosterols (e.g., β べーた campesterol , stigmasterol )Resorcylic acid lactones (e.g., zearalanone , α あるふぁ β べーた zearalenone , zeranol (α あるふぁ , taleranol (teranol, β べーた )Steroid -like (e.g., deoxymiroestrol , miroestrol )Stilbenoids (e.g., resveratrol , rhaponticin )Synthetic xenoestrogens (e.g., alkylphenols , bisphenols (e.g., BPA , BPF , BPS ), DDT , parabens , PBBs , PHBA , phthalates , PCBs )Others (e.g., agnuside , rotundifuran ) MixedSERMs Tooltip Selective estrogen receptor modulators ) Antagonists
Coregulator-binding modulators: ERX-11
GPER Tooltip G protein-coupled estrogen receptor
Agonists Antagonists Unknown
Alcohols Barbiturates Benzodiazepines Carbamates Flavonoids Imidazoles Kava constituentsMonoureides Neuroactive steroids Nonbenzodiazepines Phenols Piperidinediones Pyrazolopyridines Quinazolinones Volatiles /gases Others/unsorted
3-Hydroxybutanal α あるふぁ AA-29504 Alogabat Avermectins (e.g., ivermectin )Bromide compounds (e.g., lithium bromide , potassium bromide , sodium bromide )Carbamazepine Chloralose Chlormezanone Clomethiazole Darigabat DEABL Deuterated etifoxine Dihydroergolines (e.g., dihydroergocryptine , dihydroergosine , dihydroergotamine , ergoloid (dihydroergotoxine) )DS2 Efavirenz Etazepine Etifoxine Fenamates (e.g., flufenamic acid , mefenamic acid , niflumic acid , tolfenamic acid )Fluoxetine Flupirtine Hopantenic acid KRM-II-81 Lanthanum Lavender oil Lignans (e.g., 4-O-methylhonokiol , honokiol , magnolol , obovatol )Loreclezole Menthyl isovalerate (validolum) Monastrol Niacin Niacinamide Org 25,435 Phenytoin Propanidid Retigabine (ezogabine) Safranal Seproxetine Stiripentol Sulfonylalkanes (e.g., sulfonmethane (sulfonal) , tetronal , trional )Terpenoids (e.g., borneol )Topiramate Valerian constituents (e.g., isovaleric acid , isovaleramide , valerenic acid , valerenol )