二 乙 基 锌
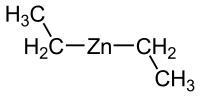
| |

| |
| IUPAC | |
| 识别 | |
| CAS |
557-20-0 |
| ChemSpider | 10413128 |
| SMILES |
|
| InChI |
|
| InChIKey | HQWPLXHWEZZGKY-GFXTWEBUAS |
| (C2H5)2Zn | |
| 123.50 g·mol⁻¹ | |
| 1.205 g/mL | |
| 熔点 | -28 °C |
| 117 °C | |
| 危险 | |
| Flammable (F); Corrosive (C); Dangerous for the environment (N) | |
合成
[编辑]安全 性
[编辑]二乙基锌和水反应非常剧烈,且在
参考 文献
[编辑]- ^ E. Frankland. On the isolation of the organic radicals. Quarterly Journal of the Chemical Society. 1850, 2 (3): 263. doi:10.1039/QJ8500200263.
- ^ Dietmar Seyferth. Zinc Alkyls, Edward Frankland, and the Beginnings of Main-Group Organometallic Chemistry. Organometallics. 2001, 20: 2940–2955. doi:10.1021/om010439f.
- ^ E. Frankland, B. F. Duppa. On a new reaction for the production of the zinc-compounds of the alkyl-radical. Journal of the Chemical Society. 1864, 17: 29–36. doi:10.1039/JS8641700029.
- ^ C. R. Noller (1943). "Diethyl Zinc". Org. Synth.; Coll. Vol. 2: 184.
外部 链接
[编辑]- Demonstration of the ignition of Diethylzinc in air Video - University of Nottingham(页面
存 档备份,存 于互联网档案 馆) - Diethylzinc, 15 wt.% solution in hexane MSDS commercial supplier(页面
存 档备份,存 于互联网档案 馆) - Diethylzinc purum MSDS commercial supplier(页面
存 档备份,存 于互联网档案 馆) - Diethylzinc MSDS commercial supplier
- Diethylzinc US MSDS commercial supplier (页面
存 档备份,存 于互联网档案 馆)
