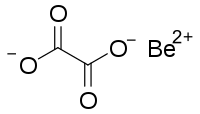
草 酸 铍
| |
| 识别 | |
| CAS |
3173-18-0(无水) 15771-43-4( 15697-24-2( |
| ChemSpider | 4953986 |
| SMILES |
|
| InChI |
|
| InChIKey | XQZGLPVUHKSNBQ-UHFFFAOYSA-L |
| C 2BeO 4 | |
| 97.03[1] g·mol⁻¹ | |
| 365.1 °C(638 K) | |
| 63.2 g( | |
| Calcium oxalate Sodium oxalate Magnesium oxalate Strontium oxalate Barium oxalate Iron(II) oxalate Iron(III) oxalate Lithium oxalate | |
2O
4,
制 备
[编辑]性 质
[编辑]参考 文献
[编辑]- ^ BERYLLIUM OXALATE. chemicalbook.com. [15 June 2021]. (
原始 内容 存 档于2022-02-04). - ^ Wirth, Fritz. Oxalates and acetates of beryllium. Zeitschrift fuer Anorganische Chemie, 1914. 87: 7-8.
- ^ Novoselova, Aleksandra Vasilʹevna; Bat︠s︡anova, Li︠u︡dmila Rafailovna. Analytical Chemistry of Beryllium. Ann Arbor-Humphrey Science Publishers. 1969: 25 [15 June 2021] (
英 语). - ^ Walsh, Kenneth A. Beryllium Chemistry and Processing. ASM International. 2009-01-01: 125 [15 June 2021]. ISBN 978-0-87170-721-5 (
英 语). - ^ Dollimore, David; Konieczay, Julie L. The thermal decomposition of beryllium oxalate and related materials. Thermochimica Acta. 1998-09-07, 318 (1–2): 155–163 [15 June 2021]. doi:10.1016/S0040-6031(98)00340-2. (
原始 内容 存 档于2022-04-01) (英 语). - ^ Moore, Raymond E. Purification of Beryllium Compounds: A Literature Survey. Oak Ridge National Laboratory. 1960: 6 [15 June 2021] (
英 语). - ^ Hamner, R. L.; Harris, L. A. Calcination in air of beryllium oxalate trihydrate to beryllium oxide. United States Atomic Energy Commission [Unclassified and Declassified Reports Published by the Atomic Energy Commission and Its Contractors], 1961. ORNL-3183.
延伸 阅读
[编辑]- Sidgwick, Nevil V.; Lewis, Neil B. CLXVI.—The solubility of beryllium oxide in solutions of its salts. J. Chem. Soc. 1926, 129 (0): 1287–1302. ISSN 0368-1769. doi:10.1039/JR9262901287.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||


![{\displaystyle {\mathsf {BeC_{2}O_{4}\cdot 3H_{2}O\ {\xrightarrow[{-2H_{2}O}]{100^{o}C}}\ BeC_{2}O_{4}\cdot H_{2}O\ {\xrightarrow[{-H_{2}O}]{220^{o}C}}\ BeC_{2}O_{4}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/26f1ccf7450ff492ba682cba56b28df05ac0b8c3)