氮化铍
| 氮化铍 | |
|---|---|
| |
| IUPAC Beryllium nitride | |
| 别名 | |
| 识别 | |
| CAS |
1304-54-7 |
| SMILES |
|
| InChI |
|
| EINECS | 215-132-6 |
| Be3N2 | |
| 55.06 g·mol⁻¹ | |
| 2.71 g/cm3[1] | |
| 熔点 | 2200 °C(2473 K)( |
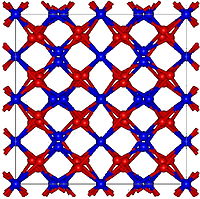
| 结构 | |
| Ia3, No. 106 ( | |
| a = 814.518 pm[2][3] | |
| 其他阳离 |
氮化钙 氮化镁 |
氮化铍,
制 备
[编辑]铍和氮在 1100 °C
用 处
[编辑]氮化铍可
性 质
[编辑]氮化铍是
它和氮化硅
氮化铍在 600 °C
参考 资料
[编辑]- ^ 1.0 1.1 1.2 1.3 1.4 1.5 Perry, Dale L. Handbook of Inorganic Compounds Second edition. Boca Raton, FL. 2011. ISBN 978-1-4398-1462-8. OCLC 759865801.
- ^ Perry, Dale L. Handbook of Inorganic Compounds Second edition. Boca Raton, FL. 2011. ISBN 978-1-4398-1462-8. OCLC 759865801.
- ^ Reckeweg, Olaf; Lind, Cora; Simon, Arndt; DiSalvo, Francis J. Rietveld Refinement of the Crystal Structure of
α -Be 3 N 2 and the Experimental Determination of Optical Band Gaps for Mg 3 N 2 , Ca 3 N 2 and CaMg 2 N 2. Zeitschrift für Naturforschung B. 2003-01-01, 58 (1): 159–162 [2021-07-21]. ISSN 1865-7117. doi:10.1515/znb-2003-0124. (原始 内容 存 档于2021-07-21) (英 语). - ^ 4.0 4.1 4.2 Egon Wiberg, Arnold Frederick Holleman (2001) Inorganic Chemistry, Elsevier ISBN 0-12-352651-5
- ^ 5.0 5.1 5.2 Ropp, R. C. Encyclopedia of the alkaline earth compounds. Oxford: Elsevier. 2013. ISBN 978-0-444-59553-9. OCLC 827243061.
- ^ Ferro, R. Intermetallic chemistry. Amsterdam: Elsevier. 2008. ISBN 978-0-08-044099-6. OCLC 281592420.
- ^ Lappin, Graham, PhD. Radiotracers in drug development. Boca Raton: CRC/Taylor & Francis. 2006. ISBN 1-4200-0498-0. OCLC 72464000.
- ^ Walsh, Kenneth A. Beryllium chemistry and processing. Materials Park, Ohio: ASM International. 2009. ISBN 978-1-61503-125-2. OCLC 770008772.
- ^ Wriedt, H. A.; Okamoto, H. The Be−N (Beryllium-Nitrogen) system. Journal of Phase Equilibria. 1987-04, 8 (2): 136–139. ISSN 1054-9714. doi:10.1007/BF02873199 (
英 语).
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||





