维基百科 ひゃっか 自由 じゆう 的 てき 百科 ひゃっか 全 ぜん
碘化亚铊 是 ぜ 一 いち 无机化合 かごう 物 ぶつ ,化学 かがく 式 しき
TlI
{\displaystyle {\ce {TlI}}}
少数 しょうすう 不可 ふか 金属 きんぞく 碘化物 ぶつ 之 これ 一 いち 包括 ほうかつ AgI 、CuI 、SnI2 、SnI4 、PbI2 和 わ HgI2 。
TlI
{\displaystyle {\ce {TlI}}}
可 か 水溶液 すいようえき 中 ちゅう 由 ゆかり 可 か 物的 ぶってき 复分解 ぶんかい 反 はん 而成。它也可 か 用 よう 乙 おつ 酸 さん 苯酚 的 てき 的 てき 形式 けいしき 出 で
所有 しょゆう 将 しょう
TlI
{\displaystyle {\ce {TlI}}}
氧化 ,产生碘化铊(
III
{\displaystyle {\ce {III}}}
的 てき 都 と 失 しつ 形成 けいせい 三 さん
Tl
+
{\displaystyle {\ce {Tl^+}}}
3 − 。
TlI
{\displaystyle {\ce {TlI}}}
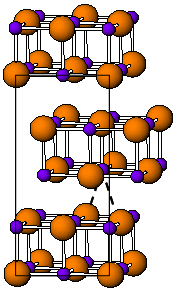
在 ざい 室温 しつおん 下 か 是 ぜ 黄色 おうしょく 的 てき 具有 ぐゆう 斜 はす 方 かた 晶 あきら 系 けい [3] 可 か 看 み 作 さく 是 ぜ 的 てき
NaCl
{\displaystyle {\ce {NaCl}}}
据 すえ 信 しんじ 的 てき 是 ぜ 由 よし 有利 ゆうり 的 てき 相互 そうご 作用 さよう 的 てき 最 さい 接近 せっきん 的 てき
Tl
−
Tl
{\displaystyle {\ce {Tl-Tl}}}
pm 。[4] 在 ざい 下 した 黄色 おうしょく 的 てき 晶 あきら 体 からだ 的 てき CsCl 晶 あきら 体 たい 相 しょう 随 ずい 着 ぎ 率 りつ 的 てき 大 だい 数量 すうりょう
TlI
{\displaystyle {\ce {TlI}}}
物 ぶつ
RbI
{\displaystyle {\ce {RbI}}}
CsI
{\displaystyle {\ce {CsI}}}
KI
{\displaystyle {\ce {KI}}}
AgI
{\displaystyle {\ce {AgI}}}
TlBr
{\displaystyle {\ce {TlBr}}}
和 わ
TlCl
{\displaystyle {\ce {TlCl}}}
可 か
CsI
{\displaystyle {\ce {CsI}}}
到 いた 室温 しつおん [5] 因 いん 在 ざい 某 ぼう 境 さかい 下 か 的 てき 存在 そんざい 可能 かのう 是 ぜ 立方 りっぽう 相 しょう 和 わ 斜 はす 方 かた 相 しょう 的 てき 相 しょう 共存 きょうぞん 的 てき 原因 げんいん [3] 在 ざい 的 てき 高 だか
TlI
{\displaystyle {\ce {TlI}}}
会 かい 金属 きんぞく 性 せい 在 ざい
LiF
{\displaystyle {\ce {LiF}}}
NaCl
{\displaystyle {\ce {NaCl}}}
或 ある
KBr
{\displaystyle {\ce {KBr}}}
上生 わぶ
TlI
{\displaystyle {\ce {TlI}}}
薄膜 うすまく 具有 ぐゆう 立方 りっぽう 石 いし [6]
碘化亚铊可 か 添加 てんか 到 いた 水 みず 光 こう 灯 とう 中 ちゅう 改善 かいぜん 性能 せいのう [7] 所 ところ 的 てき 光 ひかり 主要 しゅよう 在 ざい 可 か 青色 あおいろ 部分 ぶぶん 中 ちゅう 被 ひ 水 みず 吸收 きゅうしゅう 得 とく 最少 さいしょう 因 いん 用 よう 下 か 照明 しょうめい [8] NaI 或 ある CsI 一起 かずき 痕 こん 量 りょう 使用 しよう 射 しゃ 探 さがせ 的 てき 闪烁体 たい 探 さがせ 。
天然 てんねん 的 てき 只 ただ 是 ぜ 最近 さいきん 才 ざい 的 てき 是 ぜ 称 しょう nataliyamalikite 。它起源 げん 火山 かざん 孔 あな [9]
和 かず 化合 かごう 物 ぶつ 一 いち 有 ゆう
^ 1.0 1.1 1.2 1.3 1.4 Haynes, p. 4.94
^ Haynes, p. 4.136
^ 3.0 3.1 Lowndes, R. P.; Perry, C. H. Molecular structure and anharmonicity in thallium iodide. The Journal of Chemical Physics. 1973, 58 : 271–278. doi:10.1063/1.1678917 ^ Mudring, Anja-Verena. Thallium Halides – New Aspects of the Stereochemical Activity of Electron Lone Pairs of Heavier Main-Group Elements. European Journal of Inorganic Chemistry . 2007, 2007 (6): 882–890. doi:10.1002/ejic.200600975 ^ Sultana, Saima; Rafiuddin. Electrical conductivity in TlI–TiO2 composite solid electrolyte. Physica B: Condensed Matter. 2009, 404 : 36–40. doi:10.1016/j.physb.2008.10.002 ^ Schulz, L. G. Polymorphism of cesium and thallium halides. Acta Crystallographica. 1951, 4 (6): 487–489. doi:10.1107/S0365110X51001641 ^ Reiling, Gilbert H. Characteristics of Mercury Vapor–Metallic Iodide Arc Lamps. Journal of the Optical Society of America. 1964, 54 (4): 532. doi:10.1364/JOSA.54.000532 ^ Underwater Journal and information bulletin, IPC Science and Technology Press, (1973), p 245
^ Nataliyamalikite: Mineral information, data and localities. . www.mindat.org. [2020-09-26 ] . (原始 げんし 内容 ないよう 存 そん