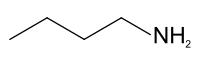
正 丁 胺
| |

| |
| IUPAC Butan-1-amine | |
| n-Butylamine | |
| 别名 |
|
| 识别 | |
| 缩写 | NBA |
| CAS |
109-73-9 |
| PubChem | 8007 |
| ChemSpider | 7716 |
| SMILES |
|
| Beilstein | 605269 |
| Gmelin | 1784 |
| UN编号 | 1125 |
| EINECS | 203-699-2 |
| ChEBI | 43799 |
| RTECS | EO29750002 |
| DrugBank | DB03659 |
| MeSH | n-butylamine |
| C4H11N | |
| 73.14 g·mol−1 | |
| 无色 | |
| 鱼味,氨味 | |
| 740 mg mL−1 | |
| 熔点 | -49 °C(224 K) |
| 77-79 °C(350-352 K) | |
| log P | 1.056 |
| 9.1 kPa ( | |
| kH | 570 |
| pKb | 3.22 |
| -58.9·10−6 cm3/mol | |
D |
1.401 |
| 黏度 | 500 µPa s( |
| 热力 | |
| −128.9–−126.5 kJ mol−1 | |
| −3.0196–−3.0174 MJ mol−1 | |
| 热容 | 188 J K−1 mol−1 |
| 危险 | |
GHS危险  
| |
| GHS |
DANGER |
| H-术语 | H225, H302, H312, H314, H332 |
| P-术语 | P210, P280, P305+351+338, P310 |
| NFPA 704 | |
| 1.7–9.8% | |
| PEL | C 5 ppm (15 mg/m3) [skin][1] |
LD50(
|
|
LCLo(
|
4000 ppm (rat, 4 hr) 263 ppm (mouse, 2 hr)[2] |
| 2- | |
制 备和反 应
[编辑]- CH3(CH2)3OH + NH3 → CH3(CH2)3NH2 + H2O
用 处
[编辑]
安全
[编辑]关于职业
参考 资料
[编辑]- ^ NIOSH Pocket Guide to Chemical Hazards. #0079. NIOSH.
- ^ 2.0 2.1 N-Butylamine. Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ H. K. Hall, Jr. Correlation of the Base Strengths of Amines. J. Am. Chem. Soc. 1957, 79 (20): 5441–5444. doi:10.1021/ja01577a030.
- ^ Rochon, Fernande D.; Buculei, Viorel. Multinuclear NMR Study and Crystal Structures of Complexes of the Types cis- and trans-Pt(amine)2I2. Inorganica Chimica Acta. 2004, 357 (8): 2218–2230. doi:10.1016/j.ica.2003.10.039.
- ^ Karsten Eller, Erhard Henkes, Roland Rossbacher, Hartmut Höke, "Amines, Aliphatic" Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005.doi:10.1002/14356007.a02_001
- ^ n-Butylamine MSDS (PDF). [2013-11-12]. (
原始 内容 (PDF)存 档于2013-11-12). - ^ CDC - NIOSH Pocket Guide to Chemical Hazards. [2021-09-11]. (
原始 内容 存 档于2021-12-17).
外部 链接
[编辑]
| |||||||||||||||||||||||||||||||||||||||||

