四 唑
| |||
| IUPAC 1H-Tetrazole | |||
| 识别 | |||
| CAS |
288-94-8 | ||
| PubChem | 67519 | ||
| ChemSpider | 60842 | ||
| SMILES |
| ||
| InChI |
| ||
| InChIKey | KJUGUADJHNHALS-UHFFFAOYAI | ||
| ChEBI | 33193 | ||
| CH2N4 | |||
| 70.05 g/mol g·mol⁻¹ | |||
| 1.477 g/mL | |||
| 熔点 | 157-158 °C [1] | ||
| 220±23 °C | |||
| pKa | -2.68 [2] | ||
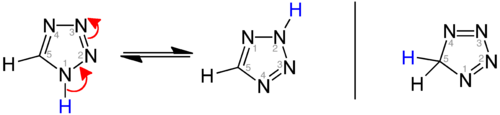
性 质
[编辑]视氢
合成
[编辑]氨基胍
应用
[编辑]1H-四唑衍生物在生物化学和制药工业上有重要应用。四唑类药物可以作为羧酸酯基的
MTT(3-(4,5-
四唑燃烧时产生高温,产物无毒,并
参考 资料
[编辑]- ^ Mihina, Joseph S.; Herbst, Robert M. The Reaction of Nitriles with Hydrazoic Acid: Synthesis of Monosubstituted Tetrazoles. J. Org. Chem. 1950, 15 (5): 1082–1092. doi:10.1021/jo01151a027.
- ^ Satchell, Jacqueline F.; Smith, Brian J. Calculation of aqueous dissociation constants of 1,2,4-triazole and tetrazole: A comparison of solvation models. Phys. Chem. Chem. Phys. 2002, 4 (18): 4314–4318. Bibcode:2002PCCP....4.4314S. doi:10.1039/b203118c.
- ^ Brockhaus ABC Chemie, VEB F. A. Brockhaus Verlag Leipzig 1965, Seite 1391.
- ^ R.A. Henry, W.G. Finnegan: An Improved Procedure for the Deamination of 5-Aminotetrazole in J. Am. Chem. Soc. 76 (1954) 290–291, doi:10.1021/ja01630a086.
- ^ F. Kurzer, L.E.A. Godfrey: Synthesen heterocyclischer Verbindungen aus Aminoguanidin in Angew. Chem. 75 (1963) 1157–1175, doi:10.1002/ange.19630752303.
| ||||||||||||||||||||||||||||||||