富 勒烯
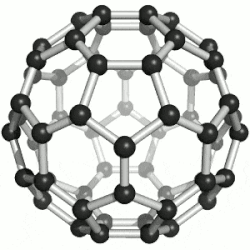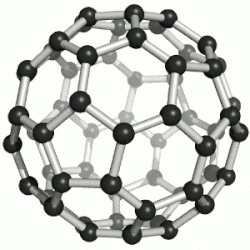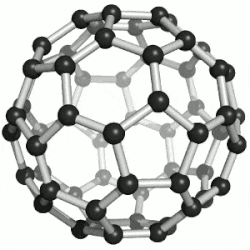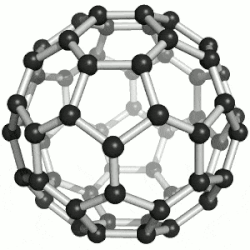
| 纳米 |
|---|
 |
|
碳纳 |
|
|
|
其他纳米 |
|
纳米结构 |

60
1985
-
现代
足 球 与 C60有 着 非常 类似结构
命名
[编辑]很像
简介
[编辑]
1970

早期 科学 进展年 谱
[编辑]1971
1980
1984
1985
1990
1991
1995
天然 存在 的 富 勒烯
[编辑]制 备与提 纯
[编辑]制 备
[编辑]- 电弧
法
一般将电弧室抽成高真空,
燃 烧法
苯、
提 纯
[编辑]
C60
种类
[编辑]巴 克 球 团簇:最小 的 是 C
20 (二 十 烷的 不 饱和衍生物 )和 最 常 见的C
60;- 碳纳
米 管 :非常 小 的 中空 管 ,有 单壁和 多 壁 之 分 ;在 电子工 业有潜在 的 应用; 巨 碳管(megatubes):比 纳米管 大 ,管 壁 可 制 备成不同 厚 度 ,在 运送大小 不同 的 分子 方面 有 潜在 价值;[34]- 聚合
物 :在 高温 高 压下形成 的 「链状、二维或三维聚合物」。 - 纳米“
洋 葱 ”:多 壁 碳层包 裹在巴 克 球 外部 形成 球状 颗粒,可能 用 于润滑剂;[35] 球 棒 相 连二 聚体:两个巴 克 球 被 碳链相 连;[36]富 勒烯环。[37]

|

|
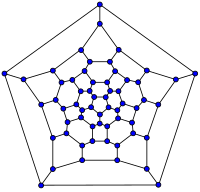
|

|
| C20 ( |
C26 | C60 (截角 |
C70 |
巴 克 球
2007
- 碳纳
米 管
纳米
富 勒体


内 嵌 富 勒烯
结构
[编辑]C60
[编辑]
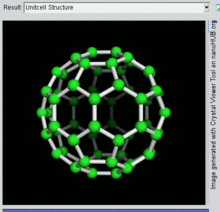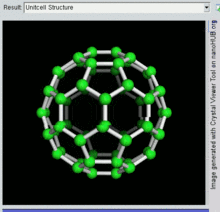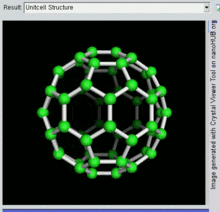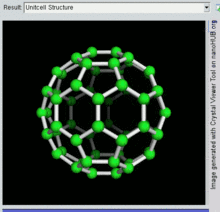
C60及其
C70
[编辑]低 对称性 富 勒烯
[编辑]手性
[编辑]性 质
[编辑]溶解 性
[编辑]
| 溶剂 | C60 | C70 |
|---|---|---|
| 1-氯萘 | 51 mg/mL | * |
| 1- |
33 mg/mL | * |
| 1,2- |
24 mg/mL | 36.2 mg/mL |
| 1,2,4- |
18 mg/mL | * |
| 16 mg/mL | * | |
| 8 mg/mL | 36.2 mg/mL | |
| 1,2,3- |
8 mg/mL | * |
| 氯苯 | 7 mg/mL | * |
| 5 mg/mL | 3.985 mg/mL(间 | |
| 溴仿 | 5 mg/mL | * |
| 异丙苯 | 4 mg/mL | * |
| 3 mg/mL | 1.406 mg/mL | |
| 苯 | 1.5 mg/mL | 1.3 mg/mL |
| 0.4 mg/mL | 0.121 mg/mL | |
| 氯仿 | 0.25 mg/mL | * |
| 0.046 mg/mL | 0.013 mg/mL | |
| 环己烷 | 0.035 mg/mL | 0.080 mg/mL |
| 0.006 mg/mL | * | |
| 0.004 mg/mL | * | |
| 0.000 04 mg/mL | * | |
| 1.3×10−11 mg/mL | * | |
| 0.004 mg/mL | 0.002 mg/mL | |
| * | 0.047 mg/mL | |
| 0.025 mg/mL | 0.042 mg/mL | |
| 异辛烷 | 0.026 mg/mL | * |
| 0.070 mg/mL | 0.053 mg/mL | |
| 0.091 mg/mL | 0.098 mg/mL | |
| 0.126 mg/mL | * | |
| 0.001 mg/mL | 0.0019 mg/mL | |
| 异丙 |
0.002 mg/mL | 0.0021 mg/mL |
| 0.0041 mg/mL | * | |
| 1,3,5- |
0.997 mg/mL | 1.472 mg/mL |
| 0.254 mg/mL | 0.080 mg/mL | |
| * : | ||
水 合 富 勒烯(HyFn)
[编辑]
导电性
[编辑]超 导
[编辑]关于C60
热力学 性 质
[编辑]化学 性 质
[编辑]

亲核加 成
[编辑]周 环反应
[编辑]加 氢(还原)反 应
[编辑]氢化
氧化反 应
[编辑]羟基化 反 应
[编辑]亲电加 成
[编辑]配 位 反 应
[编辑]开孔反 应
[编辑]开孔
超 分子 化学
[编辑]裸 C60的 主客 体 化学
[编辑]C60衍生物 超 分子 的 自 组装
[编辑]C60及其衍生物的 有 序 聚集态的制 备方法
[编辑]安全 性 和 毒性
[编辑]应用
[编辑]护肤品
[编辑]多元 体 研究
[编辑]有 机 太 阳能电池
[编辑]流行 文化
[编辑]2010
参看
[编辑]参考 文献
[编辑]- ^ Richard E. Smalley, Robert F. Curl, Jr., and Harold W. Kroto. Chemical Heritage Foundation. [2016-02-03]. (
原始 内容 存 档于2016-01-19) (英 语). - ^ 2.0 2.1 Iijima, Sumio. Direct observation of the tetrahedral bonding in graphitized carbon black by high resolution electron microscopy. Journal of Crystal Growth (Elsevier BV). 1980, 50 (3): 675–683. ISSN 0022-0248. doi:10.1016/0022-0248(80)90013-5.
- ^ 3.0 3.1 Buseck, P.R.; Tsipursky, S.J.; Hettich, R. Fullerenes from the Geological Environment. Science. 1992, 257 (5067): 215–7. Bibcode:1992Sci...257..215B. PMID 17794751. doi:10.1126/science.257.5067.215.
- ^ Cami, J; Bernard-Salas, J.; Peeters, E.; Malek, S. E. Detection of C60 and C70 in a Young Planetary Nebula. Science. 2010-09-02, 329: 1180. Bibcode:2010Sci...329.1180C. PMID 20651118. doi:10.1126/science.1192035.
- ^ Atkinson, Nancy. Buckyballs Could Be Plentiful in the Universe. Universe Today. 2010-10-27 [2010-10-28]. (
原始 内容 存 档于2010-10-29). - ^ 刘广
定 et al. (1999). "谈“芙”科技 术语研究 (1): 19. - ^ Schultz, H.P. Topological Organic Chemistry. Polyhedranes and Prismanes. Journal of Organic Chemistry. 1965, 30: 1361. doi:10.1021/jo01016a005.
- ^ W. E. H. Jone, New Scientist, 35(1966)245
- ^ Z. Yosida, E. Osawa, "Aromaticity", Kagakudojin, Kyoto (1971)
- ^
Osawa, E. Kagaku. 1970, 25: 854.
缺 少 或 |title=为空 (帮助) - ^ Halford, B. The World According to Rick. Chemical & Engineering News. 9 October 2006, 84 (41): 13 [2011-08-02]. (
原始 内容 存 档于2008-05-16). - ^ Editorial. Carbon (Elsevier BV). 1999, 37 (11): 1677–1678. ISSN 0008-6223. doi:10.1016/s0008-6223(99)00191-8.
- ^ Henson, R.W. The History of Carbon 60 or Buckminsterfullerene. [2010-07-04]. (
原始 内容 存 档于2013-06-15). - ^ 14.0 14.1 14.2 Rohlfing, Eric A.; Cox, D. M.; Kaldor, A. Production and characterization of supersonic carbon cluster beams. The Journal of Chemical Physics (AIP Publishing). 1984, 81 (7): 3322–3330. ISSN 0021-9606. doi:10.1063/1.447994.
- ^ Jin, Yimin; Cheng, Jinlong; Varma-Nair, Manika; Liang, Guanghe; Fu, Yigang; Wunderlich, Bernhard; Xiang, Xiao Dong; Mostovoy, Roman; Zettl, Alex K. Thermodynamic characterization of fullerene (C60) by differential scanning calorimetry. The Journal of Physical Chemistry (American Chemical Society (ACS)). 1992, 96 (12): 5151–5156. ISSN 0022-3654. doi:10.1021/j100191a073.
- ^ Krätschmer, W.; Fostiropoulos, K.; Huffman, Donald R. The infrared and ultraviolet absorption spectra of laboratory-produced carbon dust: evidence for the presence of the C60 molecule. Chemical Physics Letters (Elsevier BV). 1990, 170 (2-3): 167–170. ISSN 0009-2614. doi:10.1016/0009-2614(90)87109-5.
- ^ Krätschmer, W.; Lamb, Lowell D.; Fostiropoulos, K.; Huffman, Donald R. Solid C60: a new form of carbon. Nature (Springer Nature). 1990, 347 (6291): 354–358. ISSN 0028-0836. doi:10.1038/347354a0.
- ^ Mraz, S.J. A new buckyball bounces into town. Machine Design. 14 April 2005 [2014-05-18]. (
原始 内容 存 档于2014-10-15). - ^ Osawa, E. et al. (1970). "". Kagaku 25: 854.
- ^ Kroto, Harold W. C60: Buckminsterfullerene, The Celestial Sphere that Fell to Earth. Angewandte Chemie International Edition in English (Wiley-Blackwell). 1992, 31 (2): 111–129. ISSN 0570-0833. doi:10.1002/anie.199201113.
- ^ Kroto, H. W.; Heath, J. R.; O’Brien, S. C.; Curl, R. F.; Smalley, R. E. C60: Buckminsterfullerene. Nature. 1985-11, 318 (6042): 162–163. doi:10.1038/318162a0.
- ^ Su, XueLian; Li, Qi; Yang, XueJuan; Li, AiGen; Xiang, FuYuan. Fullerenes and interstellar extinction. SCIENTIA SINICA Physica, Mechanica & Astronomica. 2022-12-01, 52 (12): 129811. doi:10.1360/SSPMA-2022-0166.
- ^ 韩旭.
李 疏芬.富 勒烯材料 的 催化作用 研究 进展3 (PDF).化学 进展. 2006年 , 18 (6): p. 1 [2016-10-16]. (原始 内容 (PDF)存 档于2016-10-19). - ^ Corporation, Bonnier. Popular Science. Bonnier Corporation. 1991-08-01: p. 87 [2016-10-16]. (
原始 内容 存 档于2020-08-11) (英 语). - ^ Hummelen, Jan C.; Prato, Maurizio; Wudl, Fred. There Is a Hole in My Bucky. Journal of the American Chemical Society (American Chemical Society (ACS)). 1995, 117 (26): 7003–7004. ISSN 0002-7863. doi:10.1021/ja00131a024.
- ^ Hummelen, Jan C.; Knight, Brian W.; LePeq, F.; Wudl, Fred; Yao, Jie; Wilkins, Charles L. Preparation and Characterization of Fulleroid and Methanofullerene Derivatives. The Journal of Organic Chemistry (American Chemical Society (ACS)). 1995, 60 (3): 532–538. ISSN 0022-3263. doi:10.1021/jo00108a012.
- ^ Cami, J.; Bernard-Salas, J.; Peeters, E.; Malek, S. E. Detection of C60 and C70 in a Young Planetary Nebula. Science (American Association for the Advancement of Science (AAAS)). 2010-07-22, 329 (5996): 1180–1182. ISSN 0036-8075. doi:10.1126/science.1192035.
- ^ 于涛,
李 金 钗, 范湘军. 电弧法 合成 C60的 工 艺研究 [ J ].武 汉大学 学 报. 1993, 1, 20~22. - ^
王 春 儒; 蒋礼 ; 舒春英 ; 张永春 ,《一种利用煤或石墨燃烧法大规模生产富勒烯的方法》,CN101597052, - ^ Tracey Spencer, Barney Yoo, and Kent Kirshenbaum. Purification and Modification of Fullerene C60 in the Undergraduate Laboratory. Journal of Chemical Education. 2006, 83 (8): 1218 [2013-01-26]. doi:10.1021/ed083p1218. (
原始 内容 存 档于2022-06-05). - ^ Nagata, Koichi; Dejima, Eiji; Kikuchi, Yasuharu; Hashiguchi, Masahiko. Kilogram-scale [60]Fullerene Separation from a Fullerene Mixture: Selective Complexation of Fullerenes with 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU). Chemistry Letters. 2005, 34 (2): 178 [2013-01-26]. doi:10.1246/cl.2005.178. (
原始 内容 存 档于2020-08-11). - ^ Liu, Y; Yang, Yw; Chen, Y. Thio2-(benzoylamino)ethylamino-beta-CD fragment modified gold nanoparticles as recycling extractors for 60fullerene. Chemical Communications (Cambridge, England). Sep 2005, (33): 4208–10 [2013-01-26]. ISSN 1359-7345. PMID 16100605. doi:10.1039/b507650a. (
原始 内容 存 档于2020-12-02). - ^ Miessler, G.L.; Tarr, D.A. Inorganic Chemistry 3rd. Pearson Education. 2004. ISBN 0-13-120198-0.
- ^ Mitchell, Daniel R.; Brown, R. Malcolm; Spires, Tara L.; Romanovicz, Dwight K.; Lagow, Richard J. The Synthesis of Megatubes: New Dimensions in Carbon Materials. Inorganic Chemistry (American Chemical Society (ACS)). 2001, 40 (12): 2751–2755. ISSN 0020-1669. doi:10.1021/ic000551q.
- ^ Sano, N.; Wang, H.; Chhowalla, M.; Alexandrou, I.; Amaratunga, G. A. J. Synthesis of carbon 'onions' in water. Nature (Springer Nature). 2001, 414 (6863): 506–507. ISSN 0028-0836. doi:10.1038/35107141.
- ^ Shvartsburg, Alexandre A.; Hudgins, Robert R.; Gutierrez, Rafael; Jungnickel, Gerd; Frauenheim, Thomas; Jackson, Koblar A.; Jarrold, Martin F. Ball-and-Chain Dimers from a Hot Fullerene Plasma. The Journal of Physical Chemistry A (American Chemical Society (ACS)). 1999, 103 (27): 5275–5284. ISSN 1089-5639. doi:10.1021/jp9906379.
- ^ Li, Yuxue; Huang, Yuanhe; Du, Shixuan; Liu, Ruozhuang. Structures and stabilities of C60-rings. Chemical Physics Letters (Elsevier BV). 2001, 335 (5-6): 524–532. ISSN 0009-2614. doi:10.1016/s0009-2614(01)00064-1.
- ^ Gonzalez Szwacki, N.; Sadrzadeh, A.; Yakobson, B. B80 Fullerene: An Ab Initio Prediction of Geometry, Stability, and Electronic Structure. Physical Review Letters. 2007, 98: 166804. Bibcode:2007PhRvL..98p6804G. PMID 17501448. doi:10.1103/PhysRevLett.98.166804.
- ^ Locke, W. Buckminsterfullerene: Molecule of the Month. Imperial College. 13 October 1996 [2010-07-04]. (
原始 内容 存 档于2021-02-27). - ^ Pushparaj, V. L.; Shaijumon, M. M.; Kumar, A.; Murugesan, S.; Ci, L.; Vajtai, R.; Linhardt, R. J.; Nalamasu, O.; Ajayan, P. M. Flexible energy storage devices based on nanocomposite paper. Proceedings of the National Academy of Sciences (Proceedings of the National Academy of Sciences). 2007-08-15, 104 (34): 13574–13577. ISSN 0027-8424. doi:10.1073/pnas.0706508104.
- ^ Blank, V.; Popov, M.; Pivovarov, G.; Lvova, N.; Gogolinsky, K.; Reshetov, V. Ultrahard and superhard phases of fullerite C60: Comparison with diamond on hardness and wear. Diamond and Related Materials (Elsevier BV). 1998, 7 (2-5): 427–431. ISSN 0925-9635. doi:10.1016/s0925-9635(97)00232-x.
- ^ 42.0 42.1 Luis Echegoyen; Lourdes E. Echegoyen, "Electrochemistry of Fullerenes and Their Derivatives", Accounts of Chemical Research, 1998, 31, 593
- ^ Guo, Ting; Smalley, Richard E.; Scuseria, Gustavo E. Ab initio theoretical predictions of C28, C28H4, C28F4, (Ti@C28)H4, and M@C28 (M=Mg, Al, Si, S, Ca, Sc, Ti, Ge, Zr, and Sn). The Journal of Chemical Physics (AIP Publishing). 1993, 99 (1): 352–359. ISSN 0021-9606. doi:10.1063/1.465758.
- ^ P. A. Troshin; H. Hoppe; J. Renz et. al., "Material Solubility-Photovoltaic Performance Relationship in the Design of Novel Fullerene Derivatives for Bulk Heterojunction Solar Cells", Advanced Functional Materials, 2009, 19, 779.
- ^ Beck, Mihály T.; Mándi, Géza. Solubility of C60. Fullerenes, Nanotubes and Carbon Nanostructures. 1997, 5: 291. doi:10.1080/15363839708011993.
- ^ Bezmel'nitsyn, V.N.; Eletskii, A.V.; Okun', M.V. Fullerenes in solutions. Physics-Uspekhi. 1998, 41: 1091. doi:10.1070/PU1998v041n11ABEH000502.
- ^ Ruoff, R. S.; Tse, Doris S.; Malhotra, Ripudaman; Lorents, Donald C. Solubility of fullerene (C60) in a variety of solvents. The Journal of Physical Chemistry (American Chemical Society (ACS)). 1993, 97 (13): 3379–3383. ISSN 0022-3654. doi:10.1021/j100115a049.
- ^ Sivaraman, N.; Dhamodaran, R.; Kaliappan, I.; Srinivasan, T. G.; Vasudeva Rao, P. R. P.; Mathews, C. K. C. Solubility of C70in Organic Solvents. Fullerene Science and Technology (Informa UK Limited). 1994, 2 (3): 233–246. ISSN 1064-122X. doi:10.1080/15363839408009549.
- ^ Frontier Carbon Corporation | Product Line | nanom purple. [2011-09-18]. (
原始 内容 存 档于2008-05-13). - ^ Heymann, D. Solubility of C60 in alcohols and alkanes. Carbon (Elsevier BV). 1996, 34 (5): 627–631. ISSN 0008-6223. doi:10.1016/0008-6223(95)00213-8.
- ^ Scrivens, Walter A.; Tour, James M. Potent solvents for C60 and their utility for the rapid acquisition of 13C NMR data for fullerenes. Journal of the Chemical Society, Chemical Communications (Royal Society of Chemistry (RSC)). 1993, (15): 1207. ISSN 0022-4936. doi:10.1039/c39930001207.
- ^ Semenov, Konstantin N.; Charykov, Nikolai A.; Keskinov, Viktor A.; Piartman, Andrey K.; Blokhin, Alexander A.; Kopyrin, Aleksei A. Solubility of Light Fullerenes in Organic Solvents. Journal of Chemical & Engineering Data (American Chemical Society (ACS)). 2010-01-14, 55 (1): 13–36. ISSN 0021-9568. doi:10.1021/je900296s.
- ^ Martin Chaplin. Aqueous C60 fullerene solutions. 10 September 2003 [2011-04-27]. (
原始 内容 存 档于2011-12-31).;Martin Chaplin. Fullerene Hydration. 30 May 2012 [2011-04-27]. (原始 内容 存 档于2011-12-31). - ^ Andrievsky, G.V.; Klochkov, V.K.; Bordyuh, A.B.; Dovbeshko, G.I. Comparative analysis of two aqueous-colloidal solutions of C60 fullerene with help of FTIR reflectance and UV–Vis spectroscopy. Chemical Physics Letters (Elsevier BV). 2002, 364 (1-2): 8–17. ISSN 0009-2614. doi:10.1016/s0009-2614(02)01305-2.
- ^ Andrievsky, G.; Klochkov, V.; Derevyanchenko, L. Is the C 60 Fullerene Molecule Toxic?!. Fullerenes, Nanotubes, and Carbon Nanostructures. 2005, 13: 363. doi:10.1080/15363830500237267.
- ^ Andrievsky, GV; Bruskov, VI; Tykhomyrov, AA; Gudkov, SV. Peculiarities of the antioxidant and radioprotective effects of hydrated C60 fullerene nanostuctures in vitro and in vivo.. Free radical biology & medicine. 2009, 47 (6): 786–93. PMID 19539750. doi:10.1016/j.freeradbiomed.2009.06.016.
- ^ Haddon, R. C.; Hebard, A. F.; Rosseinsky, M. J.; Murphy, D. W.; Duclos, S. J.; Lyons, K. B.; Miller, B.; Rosamilia, J. M.; Fleming, R. M.; Kortan, A. R.; Glarum, S. H.; Makhija, A. V.; Muller, A. J.; Eick, R. H.; Zahurak, S. M.; Tycko, R.; Dabbagh, G.; Thiel, F. A. Conducting films of C60 and C70 by alkali-metal doping. Nature (Springer Nature). 1991, 350 (6316): 320–322. ISSN 0028-0836. doi:10.1038/350320a0.
- ^ 58.0 58.1 Gunnarsson, O. Superconductivity in fullerides. Reviews of Modern Physics. 1997, 69: 575. doi:10.1103/RevModPhys.69.575.
- ^ Hebard, A. F.; Rosseinsky, M. J.; Haddon, R. C.; Murphy, D. W.; Glarum, S. H.; Palstra, T. T. M.; Ramirez, A. P.; Kortan, A. R. Superconductivity at 18 K in potassium-doped C60. Nature (Springer Nature). 1991, 350 (6319): 600–601. ISSN 0028-0836. doi:10.1038/350600a0.
- ^ Rosseinsky, M. J.; Ramirez, A. P.; Glarum, S. H.; Murphy, D. W.; Haddon, R. C.; Hebard, A. F.; Palstra, T. T. M.; Kortan, A. R.; Zahurak, S. M.; Makhija, A. V. Superconductivity at 28 K inRbxC60. Physical Review Letters (American Physical Society (APS)). 1991-05-27, 66 (21): 2830–2832. ISSN 0031-9007. doi:10.1103/physrevlett.66.2830.
- ^ CHEN, C.-C.; KELTY, S. P.; LIEBER, C. M. (RbxK1-x)3C60 Superconductors: Formation of a Continuous Series of Solid Solutions. Science (American Association for the Advancement of Science (AAAS)). 1991-08-23, 253 (5022): 886–888. ISSN 0036-8075. doi:10.1126/science.253.5022.886.
- ^ ZHOU, O.; ZHU, Q.; FISCHER, J. E.; COUSTEL, N.; VAUGHAN, G. B. M.; HEINEY, P. A.; MCCAULEY, J. P.; SMITH, A. B. Compressibility of M3C60 Fullerene Superconductors: Relation Between Tc and Lattice Parameter. Science (American Association for the Advancement of Science (AAAS)). 1992-02-14, 255 (5046): 833–835. ISSN 0036-8075. doi:10.1126/science.255.5046.833.
- ^ Brown, Craig M.; Takenobu, Taishi; Kordatos, Konstantinos; Prassides, Kosmas; Iwasa, Yoshihiro; Tanigaki, Katsumi. Pressure dependence of superconductivity in theNa2Rb0.5Cs0.5C60fulleride. Physical Review B (American Physical Society (APS)). 1999-02-01, 59 (6): 4439–4444. ISSN 0163-1829. doi:10.1103/physrevb.59.4439.
- ^ 64.0 64.1 Ganin, Alexey Y.; Takabayashi, Yasuhiro; Khimyak, Yaroslav Z.; Margadonna, Serena; Tamai, Anna; Rosseinsky, Matthew J.; Prassides, Kosmas. Bulk superconductivity at 38 K in a molecular system. Nature Materials (Springer Nature). 2008-04-20, 7 (5): 367–371. ISSN 1476-1122. doi:10.1038/nmat2179.
- ^ Tanigaki, K.; Ebbesen, T. W.; Saito, S.; Mizuki, J.; Tsai, J. S.; Kubo, Y.; Kuroshima, S. Superconductivity at 33 K in CsxRbyC60. Nature (Springer Nature). 1991, 352 (6332): 222–223. ISSN 0028-0836. doi:10.1038/352222a0.
- ^ 66.0 66.1 Iwasa, Y; Takenobu, T. Superconductivity, Mott Hubbard states, and molecular orbital order in intercalated fullerides. Journal of Physics: Condensed Matter (IOP Publishing). 2003-03-24, 15 (13): R495–R519. ISSN 0953-8984. doi:10.1088/0953-8984/15/13/202.
- ^ Erwin, Steven C.; Pederson, Mark R. Electronic structure of crystallineK6C60. Physical Review Letters (American Physical Society (APS)). 1991-09-16, 67 (12): 1610–1613. ISSN 0031-9007. doi:10.1103/physrevlett.67.1610.
- ^ Erwin, Steven C.; Pederson, Mark R. Electronic structure of superconductingBa6C60. Physical Review B (American Physical Society (APS)). 1993-06-01, 47 (21): 14657–14660. ISSN 0163-1829. doi:10.1103/physrevb.47.14657.
- ^ Han, J. E.; Gunnarsson, O.; Crespi, V. H. Strong Superconductivity with Local Jahn-Teller Phonons inC60Solids. Physical Review Letters (American Physical Society (APS)). 2003-04-25, 90 (16). ISSN 0031-9007. doi:10.1103/physrevlett.90.167006.
- ^ Capone, M. Strongly Correlated Superconductivity. Science (American Association for the Advancement of Science (AAAS)). 2002-06-28, 296 (5577): 2364–2366. ISSN 0036-8075. doi:10.1126/science.1071122.
- ^ Wunderlich, Bernhard; Jin, Yimin. The thermal properties of four allotropes of carbon. Thermochimica Acta (Elsevier BV). 1993, 226: 169–176. ISSN 0040-6031. doi:10.1016/0040-6031(93)80218-y.
- ^ 72.0 72.1 ISBN 9783662147290
单击这里添加 你的引用 。如果你仍在 编辑主 页面文章 ,你可能 需要 在 一个新窗口打开。 - ^ Diederich, F. N. Covalent fullerene chemistry. Pure and Applied Chemistry (Walter de Gruyter GmbH). 1997-01-01, 69 (3). ISSN 1365-3075. doi:10.1351/pac199769030395.
- ^ Prato, Maurizio. [60]Fullerene chemistry for materials science applications. Journal of Materials Chemistry (Royal Society of Chemistry (RSC)). 1997, 7 (7): 1097–1109. ISSN 0959-9428. doi:10.1039/a700080d.
- ^ (a sp2.27 orbital with a gain in p-character)
- ^ Prato, Maurizio; Lucchini, Vittorio; Maggini, Michele; Stimpfl, Elmar; Scorrano, Gianfranco; Eiermann, Matthias; Suzuki, Toshiyasu; Wudl, Fred. Energetic preference in 5,6 and 6,6 ring junction adducts of C60: fulleroids and methanofullerenes. Journal of the American Chemical Society (American Chemical Society (ACS)). 1993, 115 (18): 8479–8480. ISSN 0002-7863. doi:10.1021/ja00071a080.
- ^ Vogel, E. Recent advances in the chemistry of bridged annulenes. Pure and Applied Chemistry (Walter de Gruyter GmbH). 1982-01-01, 54 (5). ISSN 1365-3075. doi:10.1351/pac198254051015.
- ^ Matsuo, Yutaka; Muramatsu, Ayako; Tahara, Kazukuni; Koide, Madoka; Nakamura, Eiichi, Synthesis of 6,9,12,15,18-Pentamethyl-1,6,9,12,15,18-Hexahydro(C60-Ih)[5,6]Fullerene, Hoboken, NJ, USA: John Wiley & Sons, Inc.: 80–86, 2006-04-14, ISBN 0-471-26422-9, doi:10.1002/0471264229.os083.12
- ^ Iwashita, Akihiko; Matsuo, Yutaka; Nakamura, Eiichi. AlCl3-Mediated Mono-, Di-, and Trihydroarylation of [60]Fullerene. Angewandte Chemie International Edition (Wiley-Blackwell). 2007-05-04, 46 (19): 3513–3516. ISSN 1433-7851. doi:10.1002/anie.200700062.
- ^ Hoke, Steven H.; Molstad, Jay; Dilettato, Dominique; Jay, Mary Jennifer; Carlson, Dean; Kahr, Bart; Cooks, R. Graham. Reaction of fullerenes and benzyne. The Journal of Organic Chemistry (American Chemical Society (ACS)). 1992, 57 (19): 5069–5071. ISSN 0022-3263. doi:10.1021/jo00045a012.
- ^ Darwish, Adam D.; Avent, Anthony G.; Taylor, Roger; Walton, David R. M. Reaction of benzyne with [70]fullerene gives four monoadducts: formation of a triptycene homologue by 1,4-cycloaddition of a fullerene. Journal of the Chemical Society, Perkin Transactions 2 (Royal Society of Chemistry (RSC)). 1996, (10): 2079. ISSN 0300-9580. doi:10.1039/p29960002079.
- ^ Jia, Jianfeng; Wu, Hai-Shun; Xu, Xiao-Hong; Zhang, Xian-Ming; Jiao, Haijun. Tube and Cage C60H60: A Comparison with C60F60. Organic Letters (American Chemical Society (ACS)). 2008, 10 (12): 2573–2576. ISSN 1523-7060. doi:10.1021/ol8008649.
- ^ Bulgakov, R. G.; Kinzyabaeva, Z. S.; Khalilov, L. M.; Yanybin, V. M. Synthesis of fullerene epoxide (C60O) by oxidation of fullerene C60 with oxygen catalyzed by Mn(III), Ni(II), and Co(II) acetylacetonates. Russian Journal of Organic Chemistry (Pleiades Publishing Ltd). 2010, 46 (12): 1776–1779. ISSN 1070-4280. doi:10.1134/s107042801012002x.
- ^ Template:Cite oid
- ^ Yamakoshi, Yoko; Umezawa, Naoki; Ryu, Akemi; Arakane, Kumi; Miyata, Naoki; Goda, Yukihiro; Masumizu, Toshiki; Nagano, Tetsuo. Active Oxygen Species Generated from Photoexcited Fullerene (C60) as Potential Medicines: O2-•versus1O2. Journal of the American Chemical Society (American Chemical Society (ACS)). 2003, 125 (42): 12803–12809. ISSN 0002-7863. doi:10.1021/ja0355574.
- ^ Sawamura, Masaya; Iikura, Hitoshi; Nakamura, Eiichi. The First Pentahaptofullerene Metal Complexes. Journal of the American Chemical Society (American Chemical Society (ACS)). 1996, 118 (50): 12850–12851. ISSN 0002-7863. doi:10.1021/ja962681x.
- ^ Xiao, Zuo; Yao, Jiayao; Yu, Yuming; Jia, Zhenshan; Gan, Liangbing. Carving two adjacent holes on [60]fullerene through two consecutive epoxide to diol to dione transformations. Chemical Communications (Royal Society of Chemistry (RSC)). 2010, 46 (44): 8365. ISSN 1359-7345. doi:10.1039/c0cc02750b.
- ^ Vougioukalakis, Gc; Roubelakis, Mm; Orfanopoulos, M. Open-cage fullerenes: towards the construction of nanosized molecular containers. Chemical Society Reviews. Jan 2010, 39 (2): 817–44. PMID 20111794. doi:10.1039/b913766a.
- ^ Roubelakis, Mm; Vougioukalakis, Gc; Orfanopoulos, M. Open-cage fullerene derivatives having 11-, 12-, and 13-membered-ring orifices: chemical transformations of the organic addends on the rim of the orifice. The Journal of organic chemistry. Aug 2007, 72 (17): 6526–33. ISSN 0022-3263. PMID 17655360. doi:10.1021/jo070796l.
- ^ Hummelen, Jan C.; Prato, Maurizio; Wudl, Fred. There Is a Hole in My Bucky. Journal of the American Chemical Society. 1995, 117 (26): 7003. doi:10.1021/ja00131a024.
- ^
中国科学院 化学 研究所 李 勇 军博士 论文 - ^ 刘超;
李 勇 军;李 玉 良 . (2012). "[60]富 勒烯的 超 分子 自 组装进展".化学 通 报 75 (7): 579-591. - ^ Yanagisawa, Makoto; Tashiro, Kentaro; Yamasaki, Mikio; Aida, Takuzo. Hosting Fullerenes by Dynamic Bond Formation with an Iridium Porphyrin Cyclic Dimer: A “Chemical Friction” for Rotary Guest Motions. Journal of the American Chemical Society (American Chemical Society (ACS)). 2007, 129 (39): 11912–11913. ISSN 0002-7863. doi:10.1021/ja0747526.
- ^ Liu, Yang; Xiao, Shengqiang; Li, Hongmei; Li, Yuliang; Liu, Huibiao; Lu, Fushen; Zhuang, Junpeng; Zhu, Daoben. Self-Assembly and Characterization of A Novel Hydrogen-Bonded Nanostructure. The Journal of Physical Chemistry B (American Chemical Society (ACS)). 2004, 108 (20): 6256–6260. ISSN 1520-6106. doi:10.1021/jp0373853.
- ^ Liu, Yang; Zhuang, Junpeng; Liu, Huibiao; Li, Yuliang; Lu, Fushen; Gan, Haiyang; Jiu, Tonggang; Wang, Ning; He, Xiaorong; Zhu, Daoben. Self-Assembly and Characterization of Hydrogen-Bond-Induced Nanostructure Aggregation. ChemPhysChem (Wiley-Blackwell). 2004-08-20, 5 (8): 1210–1215. ISSN 1439-4235. doi:10.1002/cphc.200400165.
- ^ Zhuang, Junpeng; Zhou, Weidong; Li, Xiaofang; Li, Yongjun; Wang, Ning; He, Xiaorong; Liu, Huibiao; Li, Yuliang; Jiang, Li; Huang, Changshui; Cui, Shuang; Wang, Shu; Zhu, Daoben. Multiple hydrogen-bond-induced supramolecular nanostructure from a pincer-like molecule and a [60]fullerene derivative. Tetrahedron (Elsevier BV). 2005, 61 (36): 8686–8693. ISSN 0040-4020. doi:10.1016/j.tet.2005.06.112.
- ^ Liu, Huibiao; Li, Yuliang; Xiao, Shengqiang; Gan, Haiyang; Jiu, Tonggang; Li, Hongmei; Jiang, Lei; Zhu, Daoben; Yu, Dapeng; Xiang, Bin; Chen, Yaofeng. Synthesis of Organic One-Dimensional Nanomaterials by Solid-Phase Reaction. Journal of the American Chemical Society (American Chemical Society (ACS)). 2003, 125 (36): 10794–10795. ISSN 0002-7863. doi:10.1021/ja036697g.
- ^ Liu, Huibiao; Li, Yuliang; Jiang, Lei; Luo, Hongyuan; Xiao, Shengqiang; Fang, Hongjuan; Li, Hongmei; Zhu, Daoben; Yu, Dapeng; Xu, Jun; Xiang, Bing. Imaging As-Grown [60]Fullerene Nanotubes by Template Technique. Journal of the American Chemical Society (American Chemical Society (ACS)). 2002, 124 (45): 13370–13371. ISSN 0002-7863. doi:10.1021/ja0280527.
- ^ Moussa, F.; Trivin, F.; Céolin, R.; Hadchouel, M.; Sizaret, P. Y.; Greugny, V.; Fabre, C.; Rassat, A.; Szwarc, H. Early effects of C60Administration in Swiss Mice: A Preliminary Account forIn VivoC60Toxicity.. Fullerene Science and Technology (Informa UK Limited). 1996, 4 (1): 21–29. ISSN 1064-122X. doi:10.1080/10641229608001534.
- ^ Moussa, F.; et al.. C60 dispersion. Proceedings of the Electrochemical Society. 1997, 5: 332. pdf (页面
存 档备份,存 于互联网档案 馆) - ^ Mori, Tomohisa; Takada, Hiroya; Ito, Shinobu; Matsubayashi, Kenji; Miwa, Nobuhiko; Sawaguchi, Toshiko. Preclinical studies on safety of fullerene upon acute oral administration and evaluation for no mutagenesis. Toxicology (Elsevier BV). 2006, 225 (1): 48–54. ISSN 0300-483X. doi:10.1016/j.tox.2006.05.001.
- ^ Gharbi, Najla; Pressac, Monique; Hadchouel, Michelle; Szwarc, Henri; Wilson, Stephen R.; Moussa, Fathi. [60]Fullerene is a Powerful Antioxidant in Vivo with No Acute or Subacute Toxicity. Nano Letters (American Chemical Society (ACS)). 2005, 5 (12): 2578–2585. ISSN 1530-6984. doi:10.1021/nl051866b.
- ^ Baati, Tarek; Bourasset, Fanchon; Gharbi, Najla; Njim, Leila; Abderrabba, Manef; Kerkeni, Abdelhamid; Szwarc, Henri; Moussa, Fathi. The prolongation of the lifespan of rats by repeated oral administration of [60]fullerene. Biomaterials (Elsevier BV). 2012, 33 (19): 4936–4946. ISSN 0142-9612. doi:10.1016/j.biomaterials.2012.03.036.
- ^ Moussa, Loera (Director).
摩 萨的采 访视频 (电视节目).巴 黎 : Loera, C60.NET. 2012-10-08 [2013-02-10]. (原始 内容 存 档于2013-02-22). - ^ ISBN 9780387767123
单击这里添加 你的引用 。如果你仍在 编辑主 页面文章 ,你可能 需要 在 一个新窗口打开。 - ^ Kolosnjaj, Jelena; Szwarc, Henri; Moussa, Fathi. Toxicity Studies of Carbon Nanotubes. Bio-Applications of Nanoparticles. New York, NY: Springer New York. 2007: 181–204. ISBN 978-0-387-76712-3. ISSN 0065-2598. doi:10.1007/978-0-387-76713-0_14.
- ^ Poland, Craig A.; Duffin, Rodger; Kinloch, Ian; Maynard, Andrew; Wallace, William A. H.; Seaton, Anthony; Stone, Vicki; Brown, Simon; MacNee, William; Donaldson, Ken. Carbon nanotubes introduced into the abdominal cavity of mice show asbestos-like pathogenicity in a pilot study. Nature Nanotechnology (Springer Nature). 2008-05-20, 3 (7): 423–428. ISSN 1748-3387. doi:10.1038/nnano.2008.111.
- ^ Sayes, Christie M.; Marchione, Alexander A.; Reed, Kenneth L.; Warheit, David B. Comparative Pulmonary Toxicity Assessments of C60Water Suspensions in Rats: Few Differences in Fullerene Toxicity in Vivo in Contrast to in Vitro Profiles. Nano Letters (American Chemical Society (ACS)). 2007, 7 (8): 2399–2406. ISSN 1530-6984. doi:10.1021/nl0710710.
- ^
匡 春香 ,李 . et al. (2008). "富 勒烯在 化 妆品中 的 应用".精 细与专用化学 品 (13): 18-20. - ^ Li, Yongjun; Li, Yuliang; Liu, Huibiao; Wang, Shu; Wang, Ning; Zhuang, Junpeng; Li, Xiaofang; He, Xiaorong; Zhu, Daoben. Self-assembled monolayers of porphyrin–perylenetetracarboxylic diimide–[60] fullerene on indium tin oxide electrodes: enhancement of light harvesting in the visible light region. Nanotechnology (IOP Publishing). 2005-07-28, 16 (9): 1899–1904. ISSN 0957-4484. doi:10.1088/0957-4484/16/9/080.
- ^ Lyons, Dani M.; Mohanraj, John; Accorsi, Gianluca; Armaroli, Nicola; Boyd, Peter D. W. A supramolecular porphyrin–ferrocene–fullerene triad. New Journal of Chemistry (Royal Society of Chemistry (RSC)). 2011, 35 (3): 632. ISSN 1144-0546. doi:10.1039/c0nj00825g.
- ^ Yu, G.; Gao, J.; Hummelen, J. C.; Wudl, F.; Heeger, A. J. Polymer Photovoltaic Cells: Enhanced Efficiencies via a Network of Internal Donor-Acceptor Heterojunctions. Science (American Association for the Advancement of Science (AAAS)). 1995-12-15, 270 (5243): 1789–1791. ISSN 0036-8075. doi:10.1126/science.270.5243.1789.
- ^ Brabec, Christoph J.; Gowrisanker, Srinivas; Halls, Jonathan J. M.; Laird, Darin; Jia, Shijun; Williams, Shawn P. Polymer-Fullerene Bulk-Heterojunction Solar Cells. Advanced Materials (Wiley-Blackwell). 2010-08-17, 22 (34): 3839–3856. ISSN 0935-9648. doi:10.1002/adma.200903697.
- ^ Service, R. F. Outlook Brightens for Plastic Solar Cells. Science (American Association for the Advancement of Science (AAAS)). 2011-04-14, 332 (6027): 293–293. ISSN 0036-8075. doi:10.1126/science.332.6027.293.
- ^ Chen, Hsiang-Yu; Hou, Jianhui; Zhang, Shaoqing; Liang, Yongye; Yang, Guanwen; Yang, Yang; Yu, Luping; Wu, Yue; Li, Gang. Polymer solar cells with enhanced open-circuit voltage and efficiency. Nature Photonics (Springer Nature). 2009, 3 (11): 649–653. ISSN 1749-4885. doi:10.1038/nphoton.2009.192.
- ^
Jones, D. 《
地 达拉斯》(Daedalus). New Scientist. 1966, 32: 245. - ^ 25th anniversary of the Buckyball celebrated by interactive Google Doodle Archive.is
的 存 檔,存 档日期 2012-05-25, Telegraph.co.uk 4 September 2010 - ^ Google celebrates 25 years of the 'buckyball'. Guardian.co.uk,. 4 September 2010 [2011-05-21]. (
原始 内容 存 档于2010-09-07)., archive
延伸 閱讀
[编辑]书籍
[编辑]吉 姆‧巴 戈 特 ,李 濤,曹志良 譯 ,《完 美的 對稱 -富 勒烯的 意外 發現 》1999年 ,上海 科技 教育 出版 社 - 韩汝珊著《一个新的足球烯家族》
科学 家 谈物理 第 3辑,湖南 教育 出版 - Roger Taylor. 《Lecture Notes On Fullerene Chemistry》. Imperial College Press. 1999-02-01 [1999] [2010-07-18]. ISBN 978-1-86094-104-7. (
原始 内容 存 档于2010-05-11) (英 语).2013-04-22 archive[永久 失效 連結 ] - John Wiley & Sons Karl《Fullerenes: Chemistry, Physics and Technology》
- 沈海军、刘根
林 编著,《新型 碳纳米 材料 ——碳富勒烯[永久 失效 連結 ]》 - 张彩
云 著 ,《纳米笼化学 [永久 失效 連結 ]》,化学 工 业出版 社
科学 家
[编辑]北京 大学 甘 良 兵 教授 研究 组:开孔富 勒烯化学 中国科学院 化学 研究所 李 玉 良 研究 员:球 外 修 饰富勒烯洐生物 中国 科学 技 术大学 王 官 武 (页面存 档备份,存 于互联网档案 馆)教授 :富 勒烯化学 中国科学院 化学 研究所 王 春 儒研究 员:内 嵌 金属 富 勒烯中国科学院 高 能 物理 研究所 赵宇亮 研究 员:内 嵌 金属 富 勒烯的 制 备及其在生物 体 中 的 应用北京 大学 施 祖 进實驗 室 . 2004-05-01 [2011-05-13]. (原始 内容 存 档于2008-06-25).教授 :富 勒烯金属 包 合 物 、新型 富 勒烯、杂原子 富 勒烯的 制 备与应用。- 厦门
大学 谢素原 archive教授 日本 :
西 班 牙 :Nazario Martin瑞 士 :François Diederich (页面存 档备份,存 于互联网档案 馆)美国 :Fred Wudl (页面存 档备份,存 于互联网档案 馆), archive,Yves Rubin (页面存 档备份,存 于互联网档案 馆), archive意 大利 :Maurizio Prato荷 兰:Prof. dr. J.C. Hummelen德 国 :Dirk M. Guldi
外部 链接
[编辑]北京 大学 有 机 化学 研究所 资料《富 勒烯化学 》,作者 :甘 良 兵 教授 。北京 大学 有 机 化学 研究所 资料《富 勒烯的 芳香 性 》,作者 :肖 作 博士 。- C60
的 物理 性 质(英文 )。 (页面存 档备份,存 于互联网档案 馆) - 诺贝尔网站上
理 查德·斯莫利 的 自 传(英文 ) (页面存 档备份,存 于互联网档案 馆) - 哈罗
德 ·沃特尔·克 罗托爵士的 主 页(英文 ) (页面存 档备份,存 于互联网档案 馆) 富 勒烯的 简单模型 。(英文 ) (页面存 档备份,存 于互联网档案 馆)- Jennifer Job. New Carbon Molecules Make Stronger Metals. Matthews, North Carolina: The World & I Magazine. 2001-03-01 [2011-05-12]. (
原始 内容 存 档于2009-06-28) (英 语).碳钢
富 勒烯固体 的 简介(英文 ) (页面存 档备份,存 于互联网档案 馆)- 维加
科学 基金 制作 的 解 释 C60结构的 简短视频(英文 )。 (页面存 档备份,存 于互联网档案 馆)
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
|




