硫代硫酸 钠
| 硫代 | |
|---|---|

| |
| |

| |
| IUPAC Sodium thiosulfate | |
| 别名 | |
| 识别 | |
| CAS |
7772-98-7 10102-17-7 |
| PubChem | 24477 |
| ChemSpider | 22885 |
| SMILES |
|
| InChI |
|
| InChIKey | AKHNMLFCWUSKQB-NUQVWONBAM |
| ChEBI | 132112 |
| RTECS | XN6476000 |
| Na 2S 2O 3 | |
| 1.667 g/cm3 | |
| 熔点 | 48.3 °C(321 K) |
| 100 °C(373 K) | |
| 70.1 g/100 mL (20 °C)[1] 231 g/100 mL (100 °C) | |
D |
1.489 |
| 结构 | |
| 危险 | |
GHS危险
| |
| GHS |
Warning |
| H-术语 | H315, H319, H335 |
| P-术语 | P261, P264, P271, P280, P302+352, P304+340, P305+351+338, P312, P321, P332+313, P337+313, P362, P403+233, P405 |
| NFPA 704 | |
| 其他阳离 |
硫代 硫代 硫代 |
硫代
硫代
用途 [编辑]
医 药用途 [编辑]
硫代
照 相 沖 印 [编辑]
溴化
自 來 水 脫 氯[编辑]
硫代
- 4 NaClO + Na
2S
2O
3 + 2 NaOH → 4 NaCl + 2 Na
2SO
4 + H
2O
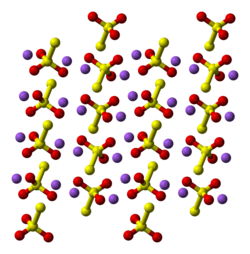
結構 [编辑]
五水合物有兩種
生產 [编辑]
- 6 NaOH + 4 S → 2 Na
2S + Na
2S
2O
3 + 3 H
2O
反應 [编辑]
- 4 Na
2S
2O
3 → 3 Na
2SO
4 + Na
2S
5
硫代
2S
2O
3(硫代
- Na
2S
2O
3 + 2 HCl → 2 NaCl + S + SO
2 + H
2O
配 位 化學 [编辑]
硫代
2O
3)
2(en)]2−,
2O
3)
2]3−
碘量法 [编辑]
- 2 S
2O2−
3 + I
2 → S
4O2−
6 + 2 I−
2S
2O
3 · 5H2O
2S
2O
3 · 5H2O也是碘鐘
這種
和 铝离子 的 反 应[编辑]
硫代
- 2 Al3+ + 3 S
2O2−
3 + 3 H
2O → 3 SO
2 + 3 S + 2 Al(OH)
3
有 机 化学 [编辑]
对硫
- ClCH
2CO
2H + Na
2S
2O
3 → Na[O
3S
2CH
2CO
2H] + NaCl - Na[O
3S
2CH
2CO
2H] + H
2O → HSCH
2CO
2H + NaHSO
4
參考 資料 [编辑]
- ^ Record in the GESTIS Substance Database from the IFA
- ^ 2.0 2.1 2.2 2.3 2.4 Sulfites, Thiosulfates, and Dithionites, Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, 2005, doi:10.1002/14356007.a25_477
- ^ 3.0 3.1 3.2 Stuart MC, Kouimtzi M, Hill SR (编). WHO Model Formulary 2008. World Health Organization. 2009: 66. ISBN 978-92-4-154765-9. hdl:10665/44053
 .
.
- ^ World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. hdl:10665/325771
 . WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. 2021. hdl:10665/345533
 . WHO/MHP/HPS/EML/2021.02.
. WHO/MHP/HPS/EML/2021.02.
- ^ Sunenshine PJ, Schwartz RA, Janniger CK. Tinea versicolor. Int. J. Dermatol. 2002, 37 (9): 648–55. PMID 9762812. S2CID 75657768. doi:10.1046/j.1365-4362.1998.00441.x.
- ^ Auriemma M, Carbone A, Di Liberato L, et al. Treatment of Cutaneous Calciphylaxis with Sodium Thiosulfate: Two Case Reports and a Review of the Literature. Am. J. Clin. Dermatol. 2011, 12 (5): 339–46. PMID 21834598. S2CID 28366905. doi:10.2165/11587060-000000000-00000.
- ^ Dickey DT, Wu YJ, Muldoon LL, et al. Protection against Cisplatin-Induced Toxicities by N-Acetylcysteine and Sodium Thiosulfate as Assessed at the Molecular, Cellular, and in Vivo Levels. J. Pharmacol. Exp. Ther. 2005, 314 (3): 1052–8. PMID 15951398. S2CID 11381393. doi:10.1124/jpet.105.087601.
- ^
國家 網 路 醫藥 , KingNet.學名 藥 SODIUM THIOSULFATE皮膚 /表皮 系統 皮膚 疾患 治療 藥品 毒物 治療 藥品 重金屬 中毒 症 |藥 典 . KingNet國家 網 路 醫藥 . [2021-05-27]. (原始 内容 存 档于2021-05-27) (中 文 ). - ^ Gibson CR. The Romance of Modern Photography, Its Discovery & Its Achievements. Seeley & Co. 1908: 37.
hyposulphite-of-soda herschel fixer hypo.
- ^ 11.0 11.1 Holleman AF, Wiberg E, Wiberg N. Inorganic Chemistry. San Diego: Academic Press. 2001. ISBN 978-0-12-352651-9.
- ^ Gordin HM. Elementary Chemistry. 1. Inorganic Chemistry. Chicago: Medico-Dental Publishing Co. 1913: 162 & 287–288.
- ^ Greenwood, Norman Neill; Earnshaw, Alan. Chemistry of the elements. 2016. ISBN 978-0-7506-3365-9. OCLC 1040112384 (
英 语). - ^ Benda, Heike von. Zur Polymorphie des Na2S2O3 / On Polymorphism of Na2S2O3. Zeitschrift für Naturforschung B (Walter de Gruyter GmbH). 1979-07-01, 34 (7): 957–968. ISSN 1865-7117. doi:10.1515/znb-1979-0716.
- ^ Aylmore MG, Muir DM. Thiosulfate Leaching of Gold - a Review. Minerals Engineering. 2001, 14 (2): 135–174. doi:10.1016/s0892-6875(00)00172-2.
- ^ Holleman, Arnold Frederik; Wiberg, Egon, Wiberg, Nils , 编, Inorganic Chemistry,
由 Eagleson, Mary; Brewer, William翻 译, San Diego/Berlin: Academic Press/De Gruyter: 594, 2001, ISBN 0-12-352651-5 - ^ Holleman, Arnold Frederik; Wiberg, Egon, Wiberg, Nils , 编, Inorganic Chemistry,
由 Eagleson, Mary; Brewer, William翻 译, San Diego/Berlin: Academic Press/De Gruyter: 670, 2001, ISBN 0-12-352651-5 - ^ Dulski TR. Ch. 8: Separation by Precypitation. A Manual for the Chemical Analysis of Metals. West Conshohocken, PA: ASTM. 1996: 99 [2021-07-30]. ISBN 9781601194350. OCLC 180851384. (
原始 内容 存 档于2021-07-30). - ^ Alonso ME, Aragona H. Sulfide Synthesis in Preparation of Unsymmetrical Dialkyl Disulfides: Sec-butyl Isopropyl Disulfide. Org. Synth. 1978, 58: 147. doi:10.15227/orgsyn.058.0147.

| ||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

