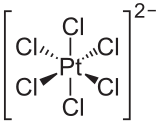铂
铂(拼音:bó,
铂
铂是
铂的应用
性 质
[编辑]物理 性 质
[编辑]纯铂为带
这些
化学 性 质
[编辑]
铂的
- Pt + 4 HNO3 + 6 HCl → H2PtCl6 + 4 NO2 + 4 H2O
铂属于软酸,
同位 素
[编辑]铂有
存 量
[编辑]

铂
1865
化合 物
[编辑]卤化物
[编辑]氯铂
氯铂
氯铂
- (H3O)2PtCl6·nH2O ⇌ PtCl4 + 2 HCl + (n + 2) H2O
- PtCl4 ⇌ PtCl2 + Cl2
- PtCl2 ⇌ Pt + Cl2
氧化物
[编辑]- 2 Pt2+ + Pt4+ + 4 O2− → Pt3O4
其他化合 物
[编辑]
3Pt
2
2Pt。[34]铂化铯(Cs
2Pt)为深红色
离子。[36]
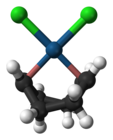
蔡斯盐
顺铂,
-
六氯铂酸离子
-
蔡斯盐阴离子
-
(1,5-环辛
二 烯)二 氯化铂(II) -
顺铂
历史
[编辑]

1741
1750
卡尔·冯·
1786
生 产
[编辑]

如果
应用
[编辑]
2010
顺铂对
此外,顺铂还用于俄歇治疗。[53]
催化剂
[编辑]铂的
度量衡 标准
[编辑]
标准铂电阻
贵金属
[编辑]
铂是贵

其他用途
[编辑]尊 贵的象 征
[编辑]
铂极为
健康 安全
[编辑]铂基
铂是
参考 资料
[编辑]- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. Standard atomic weights of the elements 2021 (IUPAC Technical Report). Pure and Applied Chemistry. 2022-05-04. ISSN 1365-3075. doi:10.1515/pac-2019-0603 (
英 语). - ^ 2.0 2.1 Braun, Mihály; Georgiev, Yordan M.; Schönherr, Tommy; Wilsenach, Heinrich; Zuber, Kai. A new precision measurement of the
α -decay half-life of 190 Pt. Physics Letters B (Elsevier BV). 2017, 768: 317–320. ISSN 0370-2693. doi:10.1016/j.physletb.2017.02.052. - ^ crustal abundance of elements. A Dictionary of Earth Sciences. Encyclopedia.com. [2017-05-21]. (
原始 内容 存 档于2017-09-23).Other elements, including such desired metals as gold (Au), silver (Ag), and platinum (Pt), are rare in the crust.
- ^ Kenneth Barbalace. Periodic Table of Elements: Platinum - Pt. EnvironmentalChemistry.com. [2017-05-21].
Earth's Crust/p.p.m.: 0.001
- ^ Activity series. cod.edu. [2017-05-21]. (
原始 内容 存 档于2015-04-29).highly unreactive
- ^ Air Quality Guidelines – Second Edition (PDF). WHO Regional Office for Europe, Copenhagen, Denmark. 2000 [2014-11-30]. (
原始 内容 存 档 (PDF)于2012-10-18). - ^ Wheate, NJ; Walker, S; Craig, GE; Oun, R. The status of platinum anticancer drugs in the clinic and in clinical trials. Dalton transactions (Cambridge, England : 2003). 2010, 39 (35): 8113–27. PMID 20593091. doi:10.1039/C0DT00292E.
- ^ 8.0 8.1 Lagowski, J. J. (编). Chemistry Foundations and Applications 3. Thomson Gale. 2004: 267–268. ISBN 0-02-865724-1.
- ^ CRC press encyclopedia of materials and finishes, 2nd edition, Mel Schwartz , 2002
- ^ Materials handbook, fifteenth edition, McGraw-Hill, by John Vaccari, 2002
- ^ 11.0 11.1 11.2 CRC contributors. Platinum. Lide, David R. (编). CRC Handbook of Chemistry and Physics 4. New York: CRC Press. 2007–2008: 26. ISBN 978-0-8493-0488-0.
- ^ Craig, Bruce D; Anderson, David S; International, A.S.M. Platinum. Handbook of corrosion data. January 1995: 8–9 [2014-11-30]. ISBN 978-0-87170-518-1. (
原始 内容 存 档于2013-12-08). - ^ 13.0 13.1 Kauffman, George B.; Thurner, Joseph J.; Zatko, David A. Ammonium Hexachloroplatinate(IV). Inorganic Syntheses. Inorganic Syntheses. 1967, 9: 182–185. ISBN 978-0-470-13240-1. doi:10.1002/9780470132401.ch51.
- ^ 14.0 14.1 14.2 Han, Y.; Huynh, H. V.; Tan, G. K. Mono- vs Bis(carbene) Complexes: A Detailed Study on Platinum(II)−Benzimidazolin-2-ylidenes. Organometallics. 2007, 26 (18): 4612. doi:10.1021/om700543p.
- ^ Audi, G.; Bersillon, O.; Blachot, J.; Wapstra, A.H. The NUBASE Evaluation of Nuclear and Decay Properties. Nuclear Physics A (Atomic Mass Data Center). 2003, 729: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001.
- ^ Earth's natural wealth: an audit (页面
存 档备份,存 于互联网档案 馆). New Scientist. May 23, 2007. - ^ Stellman, Jeanne Mager. Encyclopaedia of Occupational Health and Safety: Chemical, industries and occupations. International Labour Organization. 1998: 141 [2014-11-30]. ISBN 92-2-109816-8. (
原始 内容 存 档于2014-01-07). - ^ Murata, K. J. in Symposium on Spectrocemical Analysis for Trace Elements. ASTM International. 1958: 71 [2014-11-30]. (
原始 内容 存 档于2014-01-07). - ^ Properties, uses and production of platinum.. Canadian Platinum Corp. 2014-09-29 [2017-05-26]. (
原始 内容 存 档于2017-05-20).Sperrylite (platinum arsenide, PtAs2) ore is also a major source of this metal.
- ^ The History of Platinum. Alaska Community Database Online. ExploreNorth. [2011-04-12]. (
原始 内容 存 档于2010-12-22).Platinum is located on the Bering Sea coast, below Red Mountain on the south spit of Goodnews Bay.
- ^ Xiao, Z.; Laplante, A. R. Characterizing and recovering the platinum group minerals—a review. Minerals Engineering. 2004, 17 (9–10): 961–979. doi:10.1016/j.mineng.2004.04.001.
- ^ Dan Oancea Platinum In South Africa (页面
存 档备份,存 于互联网档案 馆). MINING.com. September 2008 - ^ 23.0 23.1 Seymour, R. J.; O'Farrelly, J. I. Platinum-group metals. Kirk Othmer Encyclopedia of Chemical Technology. Wiley. 2001. doi:10.1002/0471238961.1612012019052513.a01.pub2.
- ^ Mining Platinum in Montana. New York Times. 1998-08-13 [2008-09-09]. (
原始 内容 存 档于2008-02-03). - ^ Loferski, P. J. Platinum–Group Metals (PDF). USGS Mineral Resources Program. July 2012 [2012-07-17]. (
原始 内容 存 档 (PDF)于2017-05-13). - ^ Evidence of huge deposits of platinum in State. The Hindu (Chennai, India). 2010-07-02 [2014-11-30]. (
原始 内容 存 档于2011-12-06). - ^ MOU for platinum exploration in Tamil Nadu signed (页面
存 档备份,存 于互联网档案 馆). thaindian.com. July 1, 2010 - ^ Koeberl, Christian. Identification of meteoritic components in imactites. Meteorites: flux with time and impact effects. 1998: 133–155 [2014-11-30]. ISBN 978-1-86239-017-1. (
原始 内容 存 档于2014-04-13). - ^ 29.0 29.1 29.2 Krebs, Robert E. Platinum. The History and Use of our Earth's Chemical Elements. Greenwood Press. 1998: 124–127. ISBN 0-313-30123-9.
- ^ Smith, G. F.; Gring, J. L. The Separation and Determination of the Alkali Metals Using Perchloric Acid. V. Perchloric Acid and Chloroplatinic Acid in the Determination of Small Amounts of Potassium in the Presence of Large Amounts of Sodium. Journal of the American Chemical Society. 1933, 55 (10): 3957–3961. doi:10.1021/ja01337a007.
- ^ Schweizer, A. E.; Kerr, G. T. Thermal Decomposition of Hexachloroplatinic Acid. Inorganic Chemistry. 1978, 17 (8): 2326–2327. doi:10.1021/ic50186a067.
- ^ Perry, D. L. Handbook of Inorganic Compounds. CRC Press. 1995: 296–298. ISBN 0-8493-8671-3.
- ^ Ahrens, Sebastian; Strassner, Thomas. Detour-free synthesis of platinum-bis-NHC chloride complexes, their structure and catalytic activity in the CH activation of methane. Inorganica Chimica Acta. 2006, 359 (15): 4789. doi:10.1016/j.ica.2006.05.042.
- ^ Karpov, Andrey; Konuma, Mitsuharu; Jansen, Martin. An experimental proof for negative oxidation states of platinum: ESCA-measurements on barium platinides. Chemical Communications. 2006, (8): 838–840. PMID 16479284. doi:10.1039/b514631c.
- ^ Karpov, Andrey; Nuss, Jürgen; Wedig, Ulrich; Jansen, Martin. Cs2Pt: A Platinide(-II) Exhibiting Complete Charge Separation. Angewandte Chemie International Edition. 2003, 42 (39): 4818. doi:10.1002/anie.200352314.
- ^ 36.0 36.1 Jansen, Martin. Effects of relativistic motion of electrons on the chemistry of gold and platinum. Solid State Sciences. 2005, 7 (12): 1464. Bibcode:2005SSSci...7.1464J. doi:10.1016/j.solidstatesciences.2005.06.015.
- ^ Ghilane, J.; Lagrost, C.; Guilloux-Viry, M.; Simonet, J.; Delamar, M.; Mangeney, C.; Hapiot, P. Spectroscopic Evidence of Platinum Negative Oxidation States at Electrochemically Reduced Surfaces. Journal of Physical Chemistry C. 2007, 111 (15): 5701. doi:10.1021/jp068879d.
- ^ Richards, A.D.; Rodger, A. Synthetic metallomolecules as agents for the control of DNA structure. Chemical Society Reviews. 2007, 36 (3): 471–483 [2014-11-30]. PMID 17325786. doi:10.1039/b609495c. (
原始 内容 存 档于2007-03-03). - ^ Ken Croswell. The Stellar Origin of Copper. KenCroswell.com. 2007-04-06 [2017-05-21]. (
原始 内容 存 档于2017-01-19). - ^ Joel Achenbach. Origin of gold is likely in rare neutron-star collisions. The Washington Post. 2013-07-17 [2017-05-21]. (
原始 内容 存 档于2019-07-01). - ^ Meg Urry. Gold comes from stars. CNN. 2013-07-20 [2017-05-21]. (
原始 内容 存 档于2017-07-22). - ^ Rayner W. Hesse. Jewelrymaking Through History: An Encyclopedia. Greenwood Publishing Group. 2007: 155–156. ISBN 0-313-33507-9.
- ^ EraGem. The History of Platinum Jewelry. All Things Jewelry, Jewelry History. EraGem Post. 2013-12-02 [2017-05-21]. (
原始 内容 存 档于2024-04-02).One group of natives (La Tolita Indians) to the platinum-rich border between Ecuador and Colombia used platinum for nose rings, earrings, and masks.
- ^ 44.0 44.1 44.2 44.3 44.4 Weeks, M. E. Discovery of the Elements 7. Journal of Chemical Education. 1968: 385–407. ISBN 0-8486-8579-2. OCLC 23991202.
- ^ Dixon, Joshua; Brownrigg, William. The literary life of William Brownrigg. To which are added an account of the coal mines near Whitehaven: And Observations on the means of preventing epidemic fevers. 1801: 52 [2014-11-30]. (
原始 内容 存 档于2014-11-12). - ^ Watson, Wm; Brownrigg, William. Several Papers concerning a New Semi-Metal, Called Platina; Communicated to the Royal Society by Mr. Wm. Watson F. R. S (PDF). Philosophical Transactions. 1749, 46 (491–496): 584–596. doi:10.1098/rstl.1749.0110. (
原始 内容 (PDF)存 档于2013-10-21). - ^ Marggraf, Andreas Sigismund. Versuche mit dem neuen mineralischen Körper Platina del pinto genannt. 1760 [2014-11-30]. (
原始 内容 存 档于2015-02-24). - ^ F. w. Gibbs. William Lewis and Platina - Bicentenary of the 'Commercium Philosophico- technicum'. The Royal Institute of Chemistry, London. [2017-05-19]. (
原始 内容 存 档于2017-02-02). - ^ Platinum (页面
存 档备份,存 于互联网档案 馆). mysite.du.edu - ^ Ertl, Gerhard. Reactions at Surfaces: From Atoms to Complexity (Nobel Lecture). Angewandte Chemie International Edition. 2008, 47 (19): 385–407. PMID 18357601. doi:10.1002/anie.200800480.
- ^ 21.09kg Pt. WolframAlpha. [2012-07-14]. (
原始 内容 存 档于2014-08-23). - ^ Kelly, Thomas D. and Matos, Grecia R. (2013) Historical Statistics for Mineral and Material Commodities in the United States 互联网档
案 馆的 存 档,存 档日期 2013-06-04., U.S. Geological Survey - ^ 53.0 53.1 Loferski, P. J. 2010 Minerals Yearbook; Platinum-group metals (PDF). USGS Mineral Resources Program. October 2011 [2012-07-17]. (
原始 内容 存 档 (PDF)于2012-07-08). - ^ Heiserman, David L. Exploring Chemical Elements and their Compounds. TAB Books. 1992: 272–274. ISBN 0-8306-3018-X.
- ^ Hunt, L. B.; Lever, F. M. Platinum Metals: A Survey of Productive Resources to industrial Uses (PDF). Platinum Metals Review. 1969, 13 (4): 126–138 [2014-11-30]. (
原始 内容 (PDF)存 档于2015-09-24). - ^ Kauffman, George B.; Teter, Larry A. and Rhoda, Richard N. Recovery of Platinum from Laboratory Residues. Inorg. Synth. Inorganic Syntheses. 1963, 7: 232. ISBN 9780470132388. doi:10.1002/9780470132388.ch61.
- ^ Petrucci, Ralph H. General Chemistry: Principles & Modern Applications 9th. Prentice Hall. 2007: 606. ISBN 0-13-149330-2.
- ^ Gupta, S. V. Chapter 4. Metre Convention and Evolution of Base Units. Springer Series in Materials Science, Volume 122. 2010: 47. doi:10.1007/978-3-642-00738-5_4.
- ^ Guide tothe Realization of the ITS-90 - Platinum Resistance Thermometry (PDF). International Committee for Weights and Measures. [2020-10-23]. (
原始 内容 存 档 (PDF)于2021-02-24). - ^ Standard Reference Material 1750: Standard Platinum Resistance Thermometers,13.8033 K to 429.7485 K (PDF). NIST. [2020-10-23]. (
原始 内容 存 档 (PDF)于2020-10-26). - ^ Feltham, A. M.; Spiro, Michael. Platinized platinum electrodes. Chemical Reviews. 1971, 71 (2): 177. doi:10.1021/cr60270a002.
- ^ Professional Jeweler's Magazine Archives, issue of August 2004. [2011-06-19]. (
原始 内容 存 档于2011-09-28). - ^ Platinum primer. Diamond Cutters International. [2011-06-18]. (
原始 内容 存 档于2011-09-27). - ^ Unknown Facts about Platinum. watches.infoniac.com. [2008-09-09]. (
原始 内容 存 档于2017-11-10). - ^ Fixing Statistics. The London Platinum and Palladium Market. [2010-06-13]. (
原始 内容 存 档于2010-01-25). - ^ One Year Platinum. Kitco. [2011-06-28]. (
原始 内容 存 档于2011-09-28). - ^ Platinum versus Gold. The Speculative Invertor. 2002-04-14. (
原始 内容 存 档于2008年 10月 26日 ). - ^ Platinum. Minerals Zone. [2008-09-09]. (
原始 内容 存 档于2008年 10月 12日 ). - ^ Gwin, John. Pricing Financial Institution Products. Journal of Professional Services Marketing. 1986, 1 (3): 91. doi:10.1300/J090v01n03_07.
- ^ Crouse, Richard. Big Bang Baby: The Rock Trivia Book. 2000-05-01: 126 [2014-11-30]. ISBN 978-0-88882-219-2. (
原始 内容 存 档于2013-12-08). - ^ Gauding, Madonna. The Signs and Symbols Bible: The Definitive Guide to Mysterious Markings. 2009-10-06 [2014-11-30]. ISBN 978-1-4027-7004-3. (
原始 内容 存 档于2013-12-08). - ^ Occupational Health Guideline for Soluble Platinum Salts (as Platinum) (PDF). Centers for Disease Control and Prevention. [2008-09-09]. (
原始 内容 存 档 (PDF)于2009-06-07). - ^ FDA Backgrounder on Platinum in Silicone Breast Implants. U.S. Food and Drug Administration. [2008-09-09]. (
原始 内容 存 档于2008-07-24). - ^ Brook, Michael. Platinum in silicone breast implants. Biomaterials. 2006, 27 (17): 3274–3286. PMID 16483647. doi:10.1016/j.biomaterials.2006.01.027.
外部 链接
[编辑]元素 铂在洛 斯阿拉 莫斯国家 实验室 的 介 绍(英文 )- EnvironmentalChemistry.com —— 铂(
英文 ) 元素 铂在 The Periodic Table of Videos(诺丁汉大学 )的 介 绍(英文 )元素 铂在Peter van der Krogt elements site的 介 绍(英文 )- WebElements.com – 铂(
英文 ) - Nuclides and Isotopes (页面
存 档备份,存 于互联网档案 馆) Fourteenth Edition: Chart of the Nuclides, General Electric Company, 1989. - NIOSH Pocket Guide to Chemical Hazards – Platinum (页面
存 档备份,存 于互联网档案 馆)(NIOSH化学 危害 袖珍 指南 :铂)美国 疾病 控 制 与 预防中心 - The PGM Database. [2014-11-30]. (
原始 内容 存 档于2019-07-01). - A balanced historical account of the sequence of discoveries of platinum; illustrated.. [2014-11-30]. (
原始 内容 存 档于2010-01-23). - Platinum Metals Review: A free, quarterly journal of research on the science and technology of the platinum group metals and developments in their application in industry. [2014-11-30]. (
原始 内容 存 档于2015-05-29). - Platinum-Group Metals Statistics and Information. United States Geological Survey. [2014-11-30]. (
原始 内容 存 档于2016-02-16).
| IA 1 |
IIA 2 |
IIIB 3 |
IVB 4 |
VB 5 |
VIB 6 |
VIIB 7 |
VIIIB 8 |
VIIIB 9 |
VIIIB 10 |
IB 11 |
IIB 12 |
IIIA 13 |
IVA 14 |
VA 15 |
VIA 16 |
VIIA 17 |
VIIIA 18 | ||||||||||||||||||||
| 1 | H | He | |||||||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | |||||||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |||||||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |||||||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||||
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||