氮芥类
 |
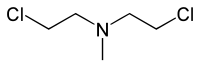 |
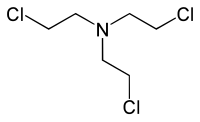 |
氮芥类物质(
虽然
氮芥类中
药物发现历史
[编辑]例 子
[编辑]- HN1:
双 (2-氯乙基 )乙 胺 - HN2:
双 (2-氯乙基 )甲 胺(氮芥) - HN3:
三 (2-氯乙基 )胺
作用 机 理
[编辑]氮介
第 一種 路 徑 :DNA中 的 親 核 性 官能 基 與 氮介類 藥物 反應 ,生成 複 合 物 ,如下圖 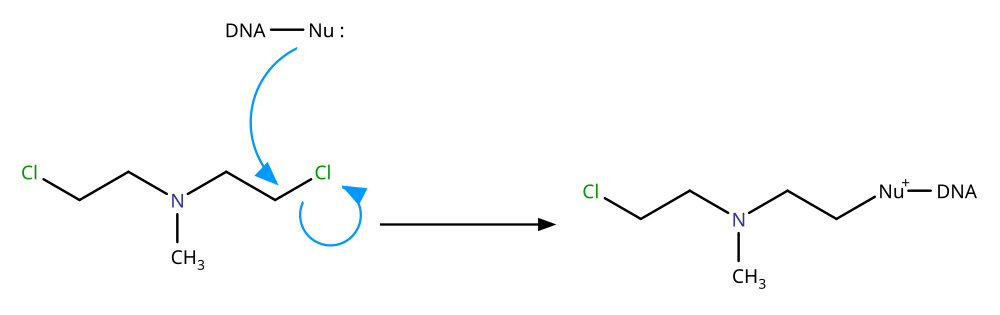
氮芥 類 藥物 第 一 條 作用 路 徑 第 二 種 路 徑 :氮芥类物质(NMs)能 够通过分子 内 缩合(脱去 Cl)形成 吖丙啶离子。吖丙啶集团随即 作 为亲电试剂被 鸟嘌呤的 亲核中心 7号 位 氮原子 进攻,发生双 分子 亲核取 代 反 应,导致鸟嘌呤的烷基化 。- 2-1
路 徑 :然 后 ,另一端发生同样的反应,在 DNA双 链间形成 交联。[6][7]一般 来 说,它们形成 的 是 在 DNA链上GNC段 5'端 的 1,3交联。[8][9][10]
氮芥类 化合 物 发生SN2反 应的机 理 - 2-2
路 徑 :另一端 發生 同樣 的 反應 ,但 是 是 與 蛋白質 中 的 親 核 性 官能 基 反應 - 2-3
路 徑 :與 單 一鳥嘌呤形成的複合物,互變成 另一構形,此構形 ,會 與 胸腺 嘧啶做鹼基 互對,導 致 DNA序列 錯誤 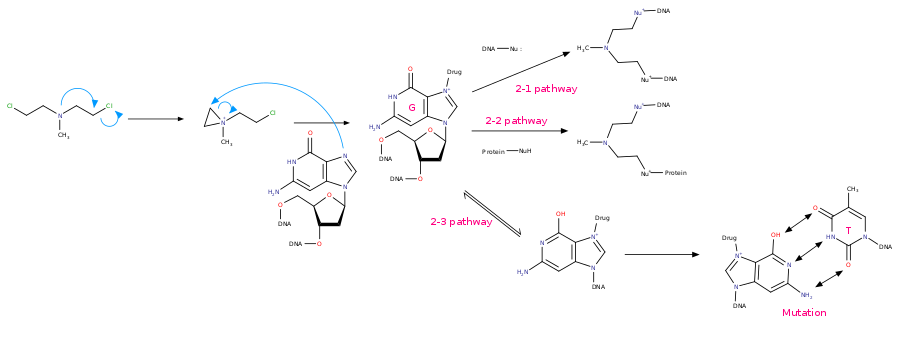
氮芥 類 藥物 第 2-1,2-2,2-3條 作用 路 徑 - 2-4
路 徑 :與 單 一鳥嘌呤形成的複合物,與 水 反應 ,使 得 嘌呤水 解 ,導 致 DNA 鏈斷裂 ,如下圖 
氮芥 類 藥物 第 2-4條 作用 路 徑 - 2-5
路 徑 :與 單 一鳥嘌呤形成的複合物,分子 內反應 使 嘌呤脫去 ,接 著 再 分子 內反應 ,使 五 碳糖開 環 ,然 後 脫 磷酸,導 致 DNA 鏈斷裂 ,如下圖 
氮芥 類 藥物 第 2-5條 作用 路 徑
- 2-1
这种对DNA
其它
参考 资料
[编辑]- ^ Gilman A. The initial clinical trial of nitrogen mustard. Am. J. Surg. May 1963, 105 (5): 574–8. PMID 13947966. doi:10.1016/0002-9610(63)90232-0.
- ^ Hirsch J. An anniversary for cancer chemotherapy. JAMA. September 2006, 296 (12): 1518–20. PMID 17003400. doi:10.1001/jama.296.12.1518.
- ^ Goodman LS, Wintrobe MM, Dameshek W, Goodman MJ, Gilman A, McLennan MT. Landmark article Sept. 21, 1946: Nitrogen mustard therapy. Use of methyl-bis(beta-chloroethyl)amine hydrochloride and tris(beta-chloroethyl)amine hydrochloride for Hodgkin's disease, lymphosarcoma, leukemia and certain allied and miscellaneous disorders. By Louis S. Goodman, Maxwell M. Wintrobe, William Dameshek, Morton J. Goodman, Alfred Gilman and Margaret T. McLennan. JAMA. May 1984, 251 (17): 2255–61. PMID 6368885. doi:10.1001/jama.251.17.2255.
- ^ Mattes, W. B.; Hartley, J. A.; Kohn, K. W. DNA sequence selectivity of guanine–N7 alkylation by nitrogen mustards. Nucleic Acids Research. 1986, 14 (7): 2971–2987. PMC 339715
 . PMID 3960738. doi:10.1093/nar/14.7.2971.
. PMID 3960738. doi:10.1093/nar/14.7.2971.
- ^ Carmen Avendano, J. Carlos Menendez. Medicinal Chemistry of Anticancer Drug. Elsevier Science. 2015.
- ^ Geiduschek EP. "Reversible" DNA. Proc. Natl. Acad. Sci. U.S.A. July 1961, 47 (7): 950–5. PMC 221307
 . PMID 13704192. doi:10.1073/pnas.47.7.950.
. PMID 13704192. doi:10.1073/pnas.47.7.950.
- ^ Brookes P, Lawley PD. The reaction of mono- and di-functional alkylating agents with nucleic acids. Biochem. J. September 1961, 80 (3): 496–503. PMC 1243259
 . PMID 16748923.
. PMID 16748923.
- ^ Rink SM, Solomon MS, Taylor MJ, Rajur SB, McLaughlin LW, Hopkins PB. Covalent structure of a nitrogen mustard-induced DNA interstrand cross-link: An N7-to-N7 linkage of deoxyguanosine residues at the duplex sequence 5'-d(GNC). Journal of the American Chemical Society. 1993, 115 (7): 2551–7. doi:10.1021/ja00060a001.
- ^ Dong Q, Barsky D, Colvin ME; et al. A structural basis for a phosphoramide mustard-induced DNA interstrand cross-link at 5'-d(GAC). Proc. Natl. Acad. Sci. U.S.A. December 1995, 92 (26): 12170–4. PMC 40318
 . PMID 8618865. doi:10.1073/pnas.92.26.12170.
. PMID 8618865. doi:10.1073/pnas.92.26.12170.
- ^ Bauer GB, Povirk LF. Specificity and kinetics of interstrand and intrastrand bifunctional alkylation by nitrogen mustards at a G-G-C sequence. Nucleic Acids Res. March 1997, 25 (6): 1211–8. PMC 146567
 . PMID 9092631. doi:10.1093/nar/25.6.1211.
. PMID 9092631. doi:10.1093/nar/25.6.1211.
- ^ Guainazzi, A.; Schärer, O. D. Using synthetic DNA interstrand crosslinks to elucidate repair pathways and identify new therapeutic targets for cancer chemotherapy. Cellular and Molecular Life Sciences. 2010, 67 (21): 3683–3697. PMID 20730555. doi:10.1007/s00018-010-0492-6.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
