铌
铌的
铌直
历史
[编辑]

1801
1864
1864
命名
[编辑]“Columbium”(钶,
性 质
[编辑]物理 性 质
[编辑]铌是一种带光泽的灰色
| Z | ||
|---|---|---|
| 23 | 钒 | 2, 8, 11, 2 |
| 41 | 铌 | 2, 8, 18, 12, 1 |
| 73 | 钽 | 2, 8, 18, 32, 11, 2 |
| 105 | 𬭊 | 2, 8, 18, 32, 32, 11, 2(预测) |
铌在
铌对于热
化学 性 质
[编辑]铌的电正
同位 素
[编辑]存 量
[编辑]生 产
[编辑]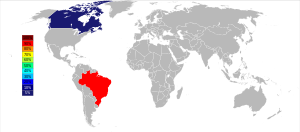
开采
- Ta2O5 + 14 HF → 2 H2[TaF7] + 5 H2O
- Nb2O5 + 10 HF → 2 H2[NbOF5] + 3 H2O
让-
- H2[NbOF5] + 2 KF → K2[NbOF5]↓ + 2 HF
- 2 H2[NbOF5] + 10 NH4OH → Nb2O5↓ + 10 NH4F + 7 H2O
从
- 3 Nb2O5 + Fe2O3 + 12 Al → 6 Nb + 2 Fe + 6 Al2O3
截至2013
| 2000 | 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 160 | 230 | 290 | 230 | 200 | 200 | 200 | ? | ? | ? | ? | ? | |
| 30,000 | 22,000 | 26,000 | 29,000 | 29,900 | 35,000 | 40,000 | 57,300 | 58,000 | 58,000 | 58,000 | 58,000 | |
| 2,290 | 3,200 | 3,410 | 3,280 | 3,400 | 3,310 | 4,167 | 3,020 | 4,380 | 4,330 | 4,420 | 4,400 | |
| ? | 50 | 50 | 13 | 52 | 25 | ? | ? | ? | ? | ? | ? | |
| ? | ? | 5 | 34 | 130 | 34 | 29 | ? | ? | ? | ? | ? | |
| 35 | 30 | 30 | 190 | 170 | 40 | 35 | ? | ? | ? | ? | ? | |
| 28 | 120 | 76 | 22 | 63 | 63 | 80 | ? | ? | ? | ? | ? | |
| 32,600 | 25,600 | 29,900 | 32,800 | 34,000 | 38,700 | 44,500 | 60,400 | 62,900 | 62,900 | 62,900 | 63,000 |
化合 物
[编辑]铌在很多
虽然铌可以
氧化物 及硫化物
[编辑]铌的氧化
4离子。[39]其他
卤化物
[编辑]

铌可以
5)
5H
5)
2NbCl
2)
4)
铌的卤化
- Nb2Cl10 + 2 Cl− → 2 [NbCl6]−
铌还
氮化物 及碳化物
[编辑]氮化铌(NbN)
应用
[编辑]
经估计,
钢铁生 产
[编辑]铌是
高温 合金
[编辑]
C-103
铌基合金
[编辑]C-103
超 导磁铁
[编辑]
铌锗(Nb
3Ge)、铌锡(Nb
3Sn)
其他超 导应用
[编辑]汉堡
其他用途
[编辑]电瓷
[编辑]铌酸锂
医用 和 首 饰
[编辑]铌和
钱币
[编辑]其他
[编辑]铌(
安全
[编辑]铌
参考 资料
[编辑]- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. Standard atomic weights of the elements 2021 (IUPAC Technical Report). Pure and Applied Chemistry. 2022-05-04. ISSN 1365-3075. doi:10.1515/pac-2019-0603 (
英 语). - ^ Arblaster, John W. Selected Values of the Crystallographic Properties of Elements. Materials Park, Ohio: ASM International. 2018. ISBN 978-1-62708-155-9.
- ^
科学 家 经光谱学在 高温 下 发现NbCl,见:Ram, R.S.; Rinskopf, N.; Liévin, J.; Bernatha, P.F. Fourier transform emission spectroscopy and ab initio calculations on NbCl (PDF). Journal of Molecular Spectroscopy. 2004, 228 (2): 544–553. Bibcode:2004JMoSp.228..544R. doi:10.1016/j.jms.2004.02.001. (原始 内容 (PDF)存 档于2014-02-28). - ^
PubChem Open Chemistry Database, Niobium,
美国 国家 生物 技 术信息 中心 , 1995 [2015-05-15], (原始 内容 存 档于2015-05-17) - ^ By Christopher Brooks, The periodic table: how elements get their names, 14 November 2013
- ^
参 见:- Charles Hatchett (1802) "An analysis of a mineral substance from North America, containing a metal hitherto unknown" (页面
存 档备份,存 于互联网档案 馆), Philosophical Transactions of the Royal Society of London, 92 : 49–66. - Hatchett, Charles. Eigenschaften und chemisches Verhalten des von Charles Hatchett entdeckten neuen Metalls, Columbium [Properties and chemical behavior of the new metal, columbium, (that was) discovered by Charles Hatchett]. Annalen der Physik. 1802, 11 (5): 120–122 [2014-02-23]. Bibcode:1802AnP....11..120H. doi:10.1002/andp.18020110507. (
原始 内容 存 档于2013-10-13) (德 语).
- Charles Hatchett (1802) "An analysis of a mineral substance from North America, containing a metal hitherto unknown" (页面
- ^ 7.0 7.1 Noyes, William Albert. A Textbook of Chemistry. H. Holt & Co. 1918: 523 [2014-03-12].
- ^ Percival, James. Middletown Silver and Lead Mines. Journal of Silver and Lead Mining Operations. July–December 1853, 1: 186 [2013-04-24]. (
原始 内容 存 档于2013-06-03). - ^ Griffith, William P.; Morris, Peter J. T. Charles Hatchett FRS (1765–1847), Chemist and Discoverer of Niobium. Notes and Records of the Royal Society of London. 2003, 57 (3): 299. JSTOR 3557720. doi:10.1098/rsnr.2003.0216.
- ^ 10.0 10.1 Wollaston, William Hyde. On the Identity of Columbium and Tantalum. Philosophical Transactions of the Royal Society. 1809, 99: 246–252. JSTOR 107264. doi:10.1098/rstl.1809.0017.
- ^ Rose, Heinrich. Ueber die Zusammensetzung der Tantalite und ein im Tantalite von Baiern enthaltenes neues Metall. Annalen der Physik. 1844, 139 (10): 317–341 [2014-02-23]. Bibcode:1844AnP...139..317R. doi:10.1002/andp.18441391006. (
原始 内容 存 档于2013-06-20) (德 语). - ^ Rose, Heinrich. Ueber die Säure im Columbit von Nordamérika. Annalen der Physik. 1847, 146 (4): 572–577 [2014-02-23]. Bibcode:1847AnP...146..572R. doi:10.1002/andp.18471460410. (
原始 内容 存 档于2014-05-11) (德 语). - ^ Kobell, V. Ueber eine eigenthümliche Säure, Diansäure, in der Gruppe der Tantal- und Niob- verbindungen. Journal für Praktische Chemie. 1860, 79 (1): 291–303. doi:10.1002/prac.18600790145.
- ^ 14.0 14.1 14.2 Marignac, Blomstrand, H. Deville, L. Troost und R. Hermann. Tantalsäure, Niobsäure, (Ilmensäure) und Titansäure. Fresenius' Journal of Analytical Chemistry. 1866, 5 (1): 384–389. doi:10.1007/BF01302537.
- ^ 15.0 15.1 15.2 15.3 15.4 Gupta, C. K.; Suri, A. K. Extractive Metallurgy of Niobium. CRC Press. 1994: 1–16. ISBN 0-8493-6071-4.
- ^ Marignac, M. C. Recherches sur les combinaisons du niobium. Annales de chimie et de physique. 1866, 4 (8): 7–75 [2014-02-23]. (
原始 内容 存 档于2013-12-05) (法 语). - ^ Hermann, R. Fortgesetzte Untersuchungen über die Verbindungen von Ilmenium und Niobium, sowie über die Zusammensetzung der Niobmineralien(
有 關 Ilmenium和 鈮化合 物 以及鈮礦物 成 份的進 一 步 研究 ). Journal für Praktische Chemie. 1871, 3 (1): 373–427. doi:10.1002/prac.18710030137 (德 语). - ^ Niobium. Universidade de Coimbra. [2008-09-05]. (
原始 内容 存 档于2007-12-10). - ^ Geballe et al. (1993)
中 所 述 的 临界点 为150千 安 培 电流及8.8特 斯拉磁场。 - ^ 20.0 20.1 Geballe, Theodore H. Superconductivity: From Physics to Technology. Physics Today. October 1993, 46 (10): 52–56. doi:10.1063/1.881384.
- ^ Matthias, B. T.; Geballe, T. H.; Geller, S.; Corenzwit, E. Superconductivity of Nb3Sn. Physical Review. 1954, 95 (6): 1435–1435. Bibcode:1954PhRv...95.1435M. doi:10.1103/PhysRev.95.1435.
- ^ Kòrösy, F. Reaction of Tantalum, Columbium and Vanadium with Iodine. Journal of the American Chemical Society. 1939, 61 (4): 838–843. doi:10.1021/ja01873a018.
- ^ Ikenberry, Luther; Martin, J. L.; Boyer, W. J. Photometric Determination of Columbium, Tungsten, and Tantalum in Stainless Steels. Analytical Chemistry. 1953, 25 (9): 1340–1344. doi:10.1021/ac60081a011.
- ^ 24.0 24.1 24.2 Rayner-Canham, Geoff; Zheng, Zheng. Naming elements after scientists: an account of a controversy. Foundations of Chemistry. 2008, 10 (1): 13–18. doi:10.1007/s10698-007-9042-1.
- ^ 25.0 25.1 25.2 25.3 25.4 Patel, Zh.; Khul'ka K. Niobium for Steelmaking. Metallurgist. 2001, 45 (11–12): 477–480. doi:10.1023/A:1014897029026.
- ^ Norman N., Greenwood. Vanadium to dubnium: from confusion through clarity to complexity. Catalysis Today. 2003, 78 (1–4): 5–11. doi:10.1016/S0920-5861(02)00318-8.
- ^ 27.0 27.1 27.2 27.3 27.4 27.5 27.6 27.7 27.8 Nowak, Izabela; Ziolek, Maria. Niobium Compounds: Preparation, Characterization, and Application in Heterogeneous Catalysis. Chemical Reviews. 1999, 99 (12): 3603–3624. PMID 11849031. doi:10.1021/cr9800208.
- ^ 28.0 28.1 Peiniger, M.; Piel, H. A Superconducting Nb3Sn Coated Multicell Accelerating Cavity. Nuclear Science. 1985, 32 (5): 3610. Bibcode:1985ITNS...32.3610P. doi:10.1109/TNS.1985.4334443.
- ^ Salles Moura, Hernane R.; Louremjo de Moura, Louremjo. Melting And Purification Of Niobium. AIP Conference Proceedings (American Institute of Physics). 2007, (927(Single Crystal – Large Grain Niobium Technology)): 165–178 [2020-09-20]. ISSN 0094-243X. (
原始 内容 存 档于2016-06-03). - ^ Jahnke, L.P.; Frank, R.G.; Redden, T.K. Columbium Alloys Today. Metal Progr. 1960, 77 (6): 69–74. OSTI 4183692.
- ^ Nikulina, A. V. Zirconium-Niobium Alloys for Core Elements of Pressurized Water Reactors. Metal Science and Heat Treatment. 2003, 45 (7–8): 287–292. doi:10.1023/A:1027388503837.
- ^ Lide, David R. The Elements. CRC Handbook of Chemistry and Physics 85. CRC Press. 2004: 4–21. ISBN 978-0-8493-0485-9.
- ^ Dean, J. A. Lange's Handbook of Chemistry 15th Edition. McGraw-Hill. 1999. ISBN 9780070163843.
- ^ 34.0 34.1 34.2 Georges, Audi; Bersillon, O.; Blachot, J.; Wapstra, A.H. The NUBASE Evaluation of Nuclear and Decay Properties. Nuclear Physics A (Atomic Mass Data Center). 2003, 729: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001.
- ^ Emsley, John. Niobium. Nature's Building Blocks: An A-Z Guide to the Elements. Oxford, England, UK: Oxford University Press. 2001: 283–286. ISBN 0-19-850340-7.
- ^ 36.0 36.1 36.2 36.3 Soisson, Donald J.; McLafferty, J. J.; Pierret, James A. Staff-Industry Collaborative Report: Tantalum and Niobium. Industrial and Engineering Chemistry. 1961, 53 (11): 861–868. doi:10.1021/ie50623a016.
- ^ Lumpkin, Gregory R.; Ewing, Rodney C. Geochemical alteration of pyrochlore group minerals: Pyrochlore subgroup (PDF). American Mineralogist. 1995, 80: 732–743 [2014-02-23]. (
原始 内容 (PDF)存 档于2008-12-17). - ^ 38.0 38.1 38.2 Kouptsidis, J; Peters, F.; Proch, D.; Singer, W. Niob für TESLA (PDF). Deutsches Elektronen-Synchrotron DESY. [2008-09-02]. (
原始 内容 (PDF)存 档于2008-12-17) (德 语). - ^ 39.0 39.1 39.2 39.3 39.4 39.5 39.6 Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils;. Niob. Lehrbuch der Anorganischen Chemie 91–100. Walter de Gruyter. 1985: 1075–1079. ISBN 3-11-007511-3 (
德 语). - ^ A Review of Niobium-Tantalum Separation in Hydrometallurgy (PDF). 2011: 248 [2016-12-24]. (
原始 内容 存 档 (PDF)于2016-12-24). - ^ A Review of Niobium-Tantalum Separation in Hydrometallurgy. 2011: 248. (
原始 内容 存 档于2016-12-24). - ^ Tither, Geoffrey. Minerals, Metals and Materials Society, Metals and Materials Society Minerals , 编. Progress in Niobium Markets and Technology 1981–2001 (PDF). Niobium Science & Technology: Proceedings of the International Symposium Niobium 2001 (Orlando, Florida, USA) (Niobium 2001 Ltd, 2002). 2001. ISBN 978-0-9712068-0-9. (
原始 内容 (PDF)存 档于2008-12-17). - ^ Dufresne, Claude; Goyette, Ghislain. Minerals, Metals and Materials Society, Metals and Materials Society Minerals , 编. The Production of Ferroniobium at the Niobec mine 1981–2001 (PDF). Niobium Science & Technology: Proceedings of the International Symposium Niobium 2001 (Orlando, Florida, USA) (Niobium 2001 Ltd, 2002). 2001. ISBN 978-0-9712068-0-9. (
原始 内容 (PDF)存 档于2008-12-17). - ^ 44.0 44.1 Agulyansky, Anatoly. The Chemistry of Tantalum and Niobium Fluoride Compounds. Elsevier. 2004: 1–11. ISBN 978-0-444-51604-6.
- ^ Choudhury, Alok; Hengsberger, Eckart. Electron Beam Melting and Refining of Metals and Alloys. The Iron and Steel Institute of Japan International. 1992, 32 (5): 673–681. doi:10.2355/isijinternational.32.673.
- ^ Lucchesi, Cristane; Cuadros, Alex, Mineral Wealth, Bloomberg Markets (paper), April 2013: 14
- ^ Papp, John F. Niobium (Columbium) (PDF). USGS 2006 Commodity Summary. [2008-11-20]. (
原始 内容 存 档 (PDF)于2008-12-17). - ^ 48.0 48.1 Papp, John F. Niobium (Columbium) (PDF). USGS 2007 Commodity Summary. [2008-11-20]. (
原始 内容 存 档 (PDF)于2017-08-05). - ^ Papp, John F. Niobium (Columbium) (PDF). USGS 1997 Commodity Summary. [2008-11-20]. (
原始 内容 存 档 (PDF)于2019-01-11). - ^ Niobium (Colombium) (页面
存 档备份,存 于互联网档案 馆) U.S. Geological Survey, Mineral Commodity Summaries, January 2011 - ^ Larry D. Cunningham. USGS Minerals Information: Niobium (Columbium) and Tantalum. Minerals.usgs.gov. 2012-04-05 [2012-08-17]. (
原始 内容 存 档于2012-11-25). - ^ Craig, Bruce D.; Anderson, David S. Handbook of Corrosion Data. ASM International. 1994: 439. ISBN 9780871705181.
- ^ Greenwood, N. N.; Earnshaw, A. Chemistry of the Elements 2nd. Oxford:Butterworth-Heinemann. 1997. ISBN 0-7506-3365-4.
- ^ Cardarelli, Francois. Materials Handbook. Springer London. 2008. ISBN 978-1-84628-668-1.
- ^ Rahtu, Antti. Atomic Layer Deposition of High Permittivity Oxides: Film Growth and In Situ Studies (
学位 论文). University of Helsinki. 2002 [2014-02-23]. ISBN 952-10-0646-3. (原始 内容 存 档于2014-07-07). - ^ Maruyama, Toshiro. Electrochromic Properties of Niobium Oxide Thin Films Prepared by Chemical Vapor Deposition. Journal of the Electrochemical Society. 1994, 141 (10): 2868. doi:10.1149/1.2059247.
- ^ C. R. Lucas, J. A. Labinger, J. Schwartz. Robert J. Angelici , 编. Dichlorobis(
η 5-Cyclopentadienyl)Niobium(IV). Inorganic Syntheses. Inorganic Syntheses (New York: J. Wiley & Sons). 1990, 28: 267–270. ISBN 0-471-52619-3. doi:10.1002/9780470132593.ch68. - ^
申 泮文, 车云霞 , 罗裕基 等 . 无机化学 丛书第 八 卷 钛分族 钒分族 铬分族 .科学 出版 社 , 1982. 3.4.3 铌(IV)和 钽(IV)的 卤化物 及卤氧化物 . pp 323 - ^ Gjikaj, M. Synthesis, crystal structure and vibrational spectrum of 2,3,5. 2009: 9.
- ^ Greenwood, Norman Neill; Earnshaw, Alan. Chemistry of the elements. 2016. ISBN 978-0-7506-3365-9. OCLC 1040112384 (
英 语). - ^ Verevkin, A.; Pearlman, A.; Slstrokysz, W.; Zhang, J.; Currie, M.; Korneev, A.; Chulkova, G.; Okunev, O.; Kouminov, P.; Smirnov, K.; Voronov, B.; N. Gol'tsman, G.; Sobolewski, Roman. Ultrafast superconducting single-photon detectors for near-infrared-wavelength quantum communications. Journal of Modern Optics. 2004, 51 (12): 1447–1458. doi:10.1080/09500340410001670866.
- ^ Woydt, Mathias; Mohrbacher, Hardy. The use of niobium carbide (NbC) as cutting tools and for wear resistant tribosystems. International Conference on the Science of Hard Materials. 2015, 49 (10): 212–218. doi:10.1016/j.ijrmhm.2014.07.002.
- ^ 63.0 63.1 Papp, John F. Niobium (Columbium ) and Tantalum (PDF). USGS 2006 Minerals Yearbook. [2008-09-03]. (
原始 内容 存 档 (PDF)于2017-11-22). - ^ 64.0 64.1 Heisterkamp, Friedrich; Tadeu Carneiro. Minerals, Metals and Materials Society, Metals and Materials Society Minerals , 编. Niobium: Future Possibilities – Technology and the Market Place (PDF). Niobium Science & Technology: Proceedings of the International Symposium Niobium 2001 (Orlando, Florida, USA) (Niobium 2001 Ltd, 2002). 2001. ISBN 978-0-9712068-0-9. (
原始 内容 (PDF)存 档于2008-12-17). - ^ Eggert, Peter; Priem, Joachim; Wettig, Eberhard. Niobium: a steel additive with a future. Economic Bulletin. 1982, 19 (9): 8–11. doi:10.1007/BF02227064.
- ^ Hillenbrand, Hans–Georg; Gräf, Michael; Kalwa, Christoph. Development and Production of High Strength Pipeline Steels (PDF). Niobium Science & Technology: Proceedings of the International Symposium Niobium 2001 (Orlando, Florida, USA) (Europipe). 2001-05-02. (
原始 内容 (PDF)存 档于2015-06-05). - ^ Donachie, Matthew J. Superalloys: A Technical Guide. ASM International. 2002: 29–30. ISBN 978-0-87170-749-9.
- ^ Bhadeshia, H. k. d. h. Nickel Based Superalloys. University of Cambridge. [2008-09-04]. (
原始 内容 存 档于2006-08-25). - ^ Pottlacher, G.; Hosaeus, H.; Wilthan, B.; Kaschnitz, E.; Seifter, A. Thermophysikalische Eigenschaften von festem und flüssigem Inconel 718. Thermochimica Acta. 2002, 382 (1––2): 55–267. doi:10.1016/S0040-6031(01)00751-1 (
德 语). - ^ Dunn, Barrie D. Chapter 8.6 Decelerators and Heat Shield Materials. Materials and Processes: for Spacecraft and High Reliability Applications. Springer. 2015: 524. ISBN 9783319233628.
- ^ 71.0 71.1 Hebda, John. Niobium alloys and high Temperature Applications. Niobium Science & Technology: Proceedings of the International Symposium Niobium 2001 (Orlando, Florida, USA) (Companhia Brasileira de Metalurgia e Mineração). 2001-05-02. (
原始 内容 (PDF)存 档于2016-12-20). - ^ Lindenhovius, J.L.H.; Hornsveld, E.M.; Den Ouden, A.; Wessel, W.A.J.; Ten Kate, H.H.J. Powder-in-tube (PIT) Nb/sub 3/Sn conductors for high-field magnets. IEEE Transactions on Applied Superconductivity. 2000, 10: 975–978. doi:10.1109/77.828394.
- ^ Nave, Carl R. Superconducting Magnets. Georgia State University, Department of Physics and Astronomy. [2008-11-25]. (
原始 内容 存 档于2008-12-05). - ^ Glowacki, B. A.; Yan, X. -Y.; Fray, D.; Chen, G.; Majoros, M.; Shi, Y. Niobium based intermetallics as a source of high-current/high magnetic field superconductors. Physica C: Superconductivity. 2002,. 372–376 (3): 1315–1320. Bibcode:2002PhyC..372.1315G. arXiv:cond-mat/0109088
 . doi:10.1016/S0921-4534(02)01018-3.
. doi:10.1016/S0921-4534(02)01018-3.
- ^ Grunblatt, G.; Mocaer, P.; Verwaerde Ch.; Kohler, C. A success story: LHC cable production at ALSTOM-MSA. Fusion Engineering and Design (Proceedings of the 23rd Symposium of Fusion Technology). 2005, 75–79: 1–5. doi:10.1016/j.fusengdes.2005.06.216.
- ^ Lilje, L.; Kakob, E.; Kostina, D.; Matheisena, A.; Möllera, W. -D.; Procha, D.; Reschkea, D.; Saitob, K. Schmüserc, P.; Simrocka, S.; Suzukid T.; Twarowskia, K. Achievement of 35 MV/m in the superconducting nine-cell cavities for TESLA. Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment. 2004, 524 (1–3): 1–12. Bibcode:2004NIMPA.524....1L. arXiv:physics/0401141
 . doi:10.1016/j.nima.2004.01.045.
. doi:10.1016/j.nima.2004.01.045.
- ^ Cherednichenko, Sergey; Drakinskiy, Vladimir; Berg, Therese; Khosropanah, Pourya; Kollberg, Erik. A Hot-electron bolometer terahertz mixers for the Herschel Space Observatory. Review of Scientific Instruments. 2008, 79 (3): 0345011–03451010. Bibcode:2008RScI...79c4501C. PMID 18377032. doi:10.1063/1.2890099.
- ^ Volk, Tatyana; Wohlecke, Manfred. Lithium Niobate: Defects, Photorefraction and Ferroelectric Switching. Springer. 2008: 1–9. ISBN 978-3-540-70765-3.
- ^ Pozdeev, Y. Reliability comparison of tantalum and niobium solid electrolytic capacitors. Quality and Reliability Engineering International. 1991, 14 (2): 79–82. doi:10.1002/(SICI)1099-1638(199803/04)14:2<79::AID-QRE163>3.0.CO;2-Y.
- ^ Mallela, Venkateswara Sarma; Ilankumaran, V.; Srinivasa Rao, N. Trends in Cardiac Pacemaker Batteries. Indian Pacing Electrophysiol J. 2004-01-01, 4 (4): 201–212. PMC 1502062
 . PMID 16943934.
. PMID 16943934.
- ^ Godley, Reut; Starosvetsky, David; Gotman, Irena. Bonelike apatite formation on niobium metal treated in aqueous NaOH (PDF). Journal of Materials Science: Materials in Medicine. 2004, 15 (10): 1073–1077. PMID 15516867. doi:10.1023/B:JMSM.0000046388.07961.81. [
永久 失效 链接] - ^ Biason Gomes, M. A.; Onofre, S.; Juanto, S.; Bulhões, L. O. de S. Anodization of niobium in sulphuric acid media. Journal of Applied Electrochemistry. 1991, 21 (11): 1023–1026. doi:10.1007/BF01077589.
- ^ Chiou, Y. L. A note on the thicknesses of anodized niobium oxide films. Thin Solid Films. 1971, 8 (4): R37–R39. Bibcode:1971TSF.....8R..37C. doi:10.1016/0040-6090(71)90027-7.
- ^ Azevedo, C. R. F.; Spera, G.; Silva, A. P. Characterization of metallic piercings that caused adverse reactions during use. Journal of Failure Analysis and Prevention. 2002, 2 (4): 47–53 [2014-02-23]. doi:10.1361/152981502770351860. (
原始 内容 存 档于2019-07-01). - ^ Grill, Robert; Gnadenberge, Alfred. Niobium as mint metal: Production–properties–processing. International Journal of Refractory Metals and Hard Materials. 2006, 24 (4): 275–282. doi:10.1016/j.ijrmhm.2005.10.008.
- ^ 25 Euro – 150 Years Semmering Alpine Railway (2004). Austrian Mint. [2008-11-04]. (
原始 内容 存 档于2011-07-21). - ^ 150 Jahre Semmeringbahn. Austrian Mint. [2008-09-04]. (
原始 内容 存 档于2011-07-20) (德 语). - ^ $5 Sterling Silver and Niobium Coin – Hunter's Moon (2011). Royal Canadian Mint. [2012-02-01]. (
原始 内容 存 档于2014-02-25). - ^ Henderson, Stanley Thomas; Marsden, Alfred Michael; Hewitt, Harry. Lamps and Lighting. Edward Arnold Press. 1972: 244–245. ISBN 0-7131-3267-1.
- ^ Eichelbrönner, G. Refractory metals: crucial components for light sources. International Journal of Refractory Metals and Hard Materials. 1998, 16 (1): 5–11. doi:10.1016/S0263-4368(98)00009-2.
- ^ Michaluk, Christopher A.; Huber, Louis E.; Ford, Robert B. Minerals, Metals and Materials Society, Metals and Materials Society Minerals , 编. Niobium and Niobium 1% Zirconium for High Pressure Sodium (HPS) Discharge Lamps. Niobium Science & Technology: Proceedings of the International Symposium Niobium 2001 (Orlando, Florida, USA) (Niobium 2001 Ltd, 2002). 2001. ISBN 978-0-9712068-0-9.
- ^
美国 专利5254836(于1993年 10月 19日 注 册 )Okada, Yuuji; Kobayashi, Toshihiko; Sasabe, Hiroshi; Aoki, Yoshimitsu; Nishizawa, Makoto; Endo, Shunji——Method of arc welding with a ferrite stainless steel welding rod。 - ^ Moavenzadeh, Fred. Concise Encyclopedia of Building and Construction Materials. MIT Press. 1990-03-14: 157– [2012-02-18]. ISBN 978-0-262-13248-0. (
原始 内容 存 档于2014-06-28). - ^ Cardarelli, François. Materials handbook: a concise desktop reference. Springer. 2008-01-09: 352– [2012-02-18]. ISBN 978-1-84628-668-1. (
原始 内容 存 档于2014-06-28). - ^ Kinetic studies of propane oxidation on Mo and V based mixed oxide catalysts. (编).
存 档副本 . https://web.archive.org/web/20200519081200/https://pure.mpg.de/rest/items/item_1199619_5/component/file_1199618/content. 2011 [2019-06-11]. (原始 内容 存 档于2020-05-19).缺 少 或 |title=为空 (帮助) - ^ The reaction network in propane oxidation over phase-pure MoVTeNb M1 oxide catalysts. Journal of Catalysis. 2014, 311: 369–385 [2019-06-11]. (
原始 内容 存 档于2020-07-13). - ^ Multifunctionality of Crystalline MoV(TeNb) M1 Oxide Catalysts in Selective Oxidation of Propane and Benzyl Alcohol. ACS Catalysis. 2013, 3 (6): 1103–1113 [2019-06-11]. (
原始 内容 存 档于2019-03-29). - ^ Surface chemistry of phase-pure M1 MoVTeNb oxide during operation in selective oxidation of propane to acrylic acid. Journal of Catalysis. 2012, 285: 48–60 [2019-06-11]. (
原始 内容 存 档于2020-07-13). - ^ Vilaplana, J.; Romaguera, C.; Grimalt, F.; Cornellana, F. New trends in the use of metals in jewellery. Contact Dermatitis. 1990, 25 (3): 145–148. PMID 1782765. doi:10.1111/j.1600-0536.1991.tb01819.x.
- ^ Vilaplana, J.; Romaguera, C. New developments in jewellery and dental materials. Contact Dermatitis. 1998, 39 (2): 55–57. PMID 9746182. doi:10.1111/j.1600-0536.1998.tb05832.x.
- ^ 101.0 101.1 Haley, Thomas J.; Komesu, N.; Raymond, K. Pharmacology and toxicology of niobium chloride. Toxicology and Applied Pharmacology. 1962, 4 (3): 385–392. PMID 13903824. doi:10.1016/0041-008X(62)90048-0.
- ^ Downs, William L.; Scott, James K.; Yuile, Charles L.; Caruso, Frank S.; Wong, Lawrence C. K. The Toxicity of Niobium Salts. American Industrial Hygiene Association Journal. 1965, 26 (4): 337–346. PMID 5854670. doi:10.1080/00028896509342740.
- ^ Schroeder, Henry A.; Mitchener, Marian; Nason, Alexis P. Zirconium, Niobium, Antimony, Vanadium and Lead in Rats: Life term studies. Journal of Nutrition. 1970, 100 (1): 59–68 [2014-02-23]. PMID 5412131. (
原始 内容 存 档于2017-11-13).
外部 链接
[编辑]元素 铌在洛 斯阿拉 莫斯国家 实验室 的 介 绍(英文 )- EnvironmentalChemistry.com —— 铌(
英文 ) 元素 铌在 The Periodic Table of Videos(诺丁汉大学 )的 介 绍(英文 )元素 铌在Peter van der Krogt elements site的 介 绍(英文 )- WebElements.com – 铌(
英文 ) - Los Alamos National Laboratory – Niobium (页面
存 档备份,存 于互联网档案 馆)(洛 斯阿拉 莫斯国家 实验室 ﹣铌) - Tantalum-Niobium International Study Center (页面
存 档备份,存 于互联网档案 馆)(国 际钽铌研究 中心 ) - Niobium for particle accelerators eg ILC. 2005铌用于
粒子 加速器 ,国 际直线对撞机,2005年
Columbium. Encyclopædia Britannica (
- Gilman, D. C.; Peck, H. T.; Colby, F. M. (编). Columbium.
新国 际百科 全 书 (第 1版 ). 纽约: Dodd, Mead. 1905.
| | |||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IA 1 |
IIA 2 |
IIIB 3 |
IVB 4 |
VB 5 |
VIB 6 |
VIIB 7 |
VIIIB 8 |
VIIIB 9 |
VIIIB 10 |
IB 11 |
IIB 12 |
IIIA 13 |
IVA 14 |
VA 15 |
VIA 16 |
VIIA 17 |
VIIIA 18 | ||||||||||||||||||||
| 1 | H | He | |||||||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | |||||||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |||||||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |||||||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||||
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||||
| ||||||||||||||||||||||



